| Posted: Aug 13, 2009 | |
DNA-encasing increases carbon nanotubes' tumor killing power |
|
| (Nanowerk Spotlight) Various forms of hyperthermia – a form of cancer treatment with elevated temperature in the range of 41-45°C – have been intensively developed for the past few decades to provide cancer clinics with more effective and advanced cancer therapy techniques. The recent use of nanomaterials has shown promising for developing more effective hyperthermia agents (read more: Self-heating nanoparticles as tumor-destroying hyperthermia agents). | |
| While most nanomedical hyperthermia research is conducted with various nanoparticles, carbon nanotubes are also of interest in these thermal ablation applications. So far, however, the utility of carbon nanotubes for in vivo use has been limited by self-association – i.e. they stick to each other. A new study has now demonstrated that DNA-encasement of multi-walled carbon nanotubes (MWCNTs) results in well-dispersed, single MWCNTs that are soluble in water and that display enhanced heat production efficiency relative to non-DNA-encased MWCNTs. | |
| "We showed that single-stranded DNA efficiently solubilizes multi-walled carbon nanotubes," William H. Gmeiner, a professor in the Department of Cancer Biology at Wake Forest University, tells Nanowerk. "Further, the DNA-encasement of the MWCNTs actually increased the amount of heat produced upon irradiation of the nanotubes with near-infrared light. Importantly, we were able to show that the heat produced by the DNA-encased MWCNTs was sufficient to selectively and completely eradicate a tumor mass without causing any significant toxicity to surrounding tissue." | |
 |
|
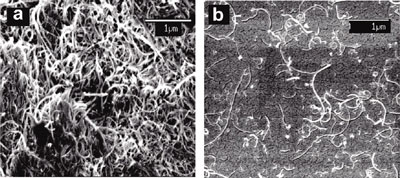
| SEM images for (left) non-DNA-encased MWCNTs and (right) DNA-encased MWCNTs. Upon DNA-encasement, the MWCNTs are well-dispersed with few aggregates observed. (Reprinted with permission from American Chemical Society) | |
| Gmeiner explains that DNA-encasement has been used previously with single-walled nanotubes (see: "Carbon nanotubes as multifunctional biological transporters and near-infrared agents for selective cancer cell destruction") and that it was shown that they may be excited upon irradiation with near-infrared light (nIR) to release heat sufficient to kill cancer cells in tissue culture. MWCNTs, though, have a larger diameter than SWCNTs and it was not known how well this strategy would work for them. It was also not known if DNA-encasement would inhibit the thermal ablative properties of MWCNTs. | |
| The Wake Forest team has now shown that it is possible to prepare DNA-encased MWCNTs and quantify the production of heat upon nIR irradiation. Their findings also indicate that control of these parameters – power and time – can be exercised to provide the desired selectivity of cell kill without damaging surrounding normal tissue. | |
| Reporting their findings in the August 5, 2009 online edition of ACS Nano ("Increased Heating Efficiency and Selective Thermal Ablation of Malignant Tissue with DNA-Encased Multiwalled Carbon Nanotubes"), Gmeiner and his team also demonstrate that the conversion of nIR irradiation to heat by DNA-encased MWCNTs is linear with respect to both power and time. | |
| Gmeiner explains that modest temperature increases of 3-5°C are sufficient to cause protein denaturation and subsequently cell death. "Our present study demonstrates that DNA-encasement of multi-walled carbon nanotubes increases heat production 2- to 3-fold relative to non-DNA-encased MWCNTs" he says. "This increased efficiency in heat production occurs over a wide range of MWCNT concentrations and irradiation times and a moderate range of power levels. Importantly, the time-dependent increase in temperature for both DNA- encased MWCNTs and non-DNA-encased MWCNTs was linear under all conditions that we evaluated." | |
| Since their SEM and AFM images clearly show that the DNA-encased MWCNTs are well-dispersed – in contrast to the non-encased nanotubes that tend to lump together – the researchers assume that decreased aggregation is the likely origin of the increased heating from DNA-encased MWCNTs relative to non-DNA-encased MWCNTs. | |
| This increased heating efficiency means that effective hyperthermia treatments could be developed using a lower concentration of carbon nanotubes – a positive development given the toxicity concerns surrounding the medical use of carbon nanotubes. Control of the power/time parameters also mean that, for a given MWCNT concentration, the required laser power could be decreased by increasing the irradiation time producing the same amount of heat while reducing damage to normal surrounding tissue in vivo. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
