| Posted: Jan 16, 2007 | |
Improving the tools for single-cell nanosurgery |
|
| (Nanowerk Spotlight) Nanosurgery holds the promise of studying or manipulating and repairing individual cells without damaging the cell. For instance, nanosurgery could remove or replace certain sections of a damaged gene inside a chromosome; sever axons to study the growth of nerve cells; or destroying an individual cell without affecting the neighboring cells. | |
| While the cell nucleus has been transplanted between cells during cloning using micropipette technologies, these methods are too crude for other subcellular structures. First steps towards nanosurgery have been made using so-called 'optical tweezers', where the energy of laser light is used to trap and manipulate nanoscale objects, for instance the nucleus of a cell, without mechanical contact. | |
| Combined with a laser scalpel (use of lasers for cutting and ablating biological objects) optical tweezers have been used to study cell fusion, DNA-cutting, etc. Unfortunately, while optical tweezers offer exquisite sensitivity in their ability to position micro- and nanoparticles, they suffer from one important disadvantage: the trapped particle is localized at the laser focus where light intensity is the highest. | |
| As a result, the laser light used to trap a particle also has a propensity to photobleach and photodamage the particle, especially when the particle is fragile and small (e.g., a subcellular organelle that is fluorescently labeled). Minimizing this drawback, new research describes the use of polarization-shaped optical vortex traps for the manipulation of particles and subcellular structures. | |
| An optical vortex, or Laguerre-Gaussian (LG) beam, is laser light that has a helical phase distribution at its wavefront. Optical vortices are characterized by the presence of a dark core, i.e. a point of zero intensity. Because of the spiral phase distribution at the wavefront, the phase at the center of the beam is undefined and thus a singularity is formed where the light intensity has a zero value. In simple terms: Light can be twisted like a corkscrew around its axis of travel. Because of the twisting, the light waves at the axis itself cancel each other out. On a flat surface, an optical vortex looks like a ring of light, with a dark hole in the center. This corkscrew of light, with darkness at the center, is called an optical vortex. | |
| "The dark core of an optical vortex takes on different appearances upon tight focusing through a high numerical aperture objective, depending on the polarization state of the incident beam" Dr. Daniel T. Chiu explains to Nanowerk. "We take advantage of these polarization effects to fashion vortex traps for the trapping of particles of different sizes while minimizing photodamage to the trapped particles and subcellular organelles." | |
| Chiu is professor of chemistry at the University of Washington. His group focuses on the development of new tools that combine ultrasensitive laser-based detection and manipulation methodologies with micro- and nano- fabrication techniques for interfacing with biological systems at the nanometer scale. His recent research was published in the December 29, 2006 web edition of Nano Letters ("Using Polarization-Shaped Optical Vortex Traps for Single-Cell Nanosurgery"). | |
| In their experiments, Chiu and his colleagues found that the photodamage is always less using a polarization-shaped vortex trap than Gaussian trap. Similarly, by comparing the time course of bleaching of mitochondria, the researchers found that the mitochondria trapped in the optical tweezer were rapidly bleached within the first few seconds, but the ones held by the vortex trap displayed a relatively stable fluorescent signal over this duration. | |
 |
|
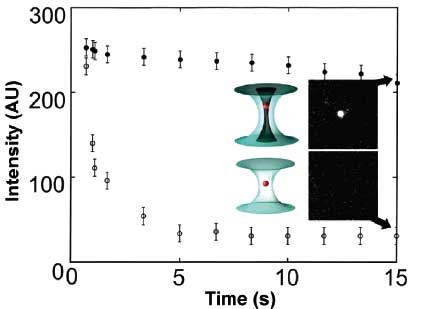
| A time plot that compares photobleaching of trapped mitochondria (stained with Mitotracker Green dye) in a conventional optical tweezer and in a linearly polarized vortex trap (laser power is 75 mW at the object plane): (o) mitochondria trapped in the optical tweezer; (•) mitochondria held in the vortex trap. To check for fluorescence, the trapped and stained mitochondria were illuminated periodically with 488 nm excitation from an Ar+ laser; the fluorescence observed under 488 nm excitation (ordinate) was plotted as a function of trapping time (abscissa). The insets show fluorescence images (obtained using 488 nm illumination) of mitochondria trapped for 15 s (arrows) in the optical tweezer and in the vortex trap. (Reprinted with permission from the American Chemical Society) | |
| Applying these findings to single-cell nanosurgery, Chiu demonstrates that the use of polarization- shaped optical vortex traps minimizes photobleaching of fluorescent tagging and damage to the cell caused by optical tweezers. | |
| "The ability to manipulate optically submicrometer subcellular structures while minimizing photodamage has several practical applications" says Chiu. "In particular, we are developing a droplet nanolab platform for single-organelle assays and chemical analysis, because many diseases are caused by organelle malfunction where a given cell can contain a heterogeneous population of healthy and defective organelles. To achieve single-organelle analysis, it requires the optical manipulation and encapsulation of the select organelle within a femtoliter-volume aqueous droplet. Here, it is pertinent the organelle remains functionally intact during optical manipulation so subsequent biological or chemical analysis reflects the true physiological state of the organelle." | |
| Chiu points out their study points to a new strategy based on shaping intensity variations within the diffraction-limited laser focus to create the next generation of optical tweezers. | |
| "The availability of such tools offers unprecedented control in active nanoscopic manipulation, from subcellular structures to nanoparticles, such as metal colloids and quantum dots" says Chiu. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
