| Posted: Jan 17, 2007 | |
Novel nanofluidic device could lead to electrical single molecule detection |
|
| (Nanowerk Spotlight) The ability to detect few or individual molecules in solution is at present largely limited to fluorescence techniques, and a comparable method using electrical detection has so far remained elusive. Such a technique would be highly desirable for lab-on-a-chip applications and when labeling with fluorophores is invasive or impossible. More importantly, it would pave the way for fluidic devices in which individual ions are electrically detected and manipulated, allowing a new class of fundamental experiments on nonequilibrium statistical physics, transport at the molecular scale, and a broad range of biophysical systems. Researchers in The Netherlands now have demonstrated a new nanofluidic device for the detection of electrochemical active molecules with an extremely high sensitivity. A prototype device allows detecting fluctuations due to Brownian motion of as few as ∼70 molecules, a level heretofore unachieved in electrochemical sensors. Ultimately, the researchers hope the device will not only be able to detect single molecules in the device, but also discriminate between various species. | |
| This nanofluidic device, developed by scientists at the Kavli Institute of Nanoscience at Delft University of Technology in The Netherlands, achieves highly sensitive electrical detection of electroactive ions through efficient redox cycling. | |
| Marcel Zevenbergen, a PhD student at the Kavli Institute's Molecular Biophysics Group, explains the novelty of the device to Nanowerk: "Electrochemical active molecules are repeatedly oxidized and reduced at two closely-spaced platinum electrodes thereby generating a current. This concept is routinely used in scanning electrochemical microscopy and single-molecule detection was achieved more than a decade ago. However, the poor reproducibility of this particular approach prevented it from evolving beyond the proof-of-principle stage. By introducing a solid-state device we have far better control over the size dimensions and we believe that this will ultimately allow to electrical detection of single molecules in solution." | |
| Zevenbergen and his colleagues demonstrate that the noise in the detection signal is directly related to the diffusive motion of molecules entering and leaving the device. This rare type of noise is called “diffusion noise” and has been partially observed in a small number of systems, but never with simple ions. | |
| "We have unambiguously observed diffusion noise because our device is the first direct experimental realization of a theoretical framework developed in the 50’s" says Zevenbergen. "Our data agrees quantitatively with this model without any fitting parameter." | |
| "Most importantly, this good agreement with theory shows that the device works exactly as we designed it" adds Dr. Serge Lemay. Lemay is one of the leaders of the Molecular Biophysics group. | |
| At the core of the device is a solution-filled cavity bounded by two parallel electrodes separated by less than 300 nm. Redox cycling permits an approx. 400-fold amplification of the electrochemical current. | |
 |
|
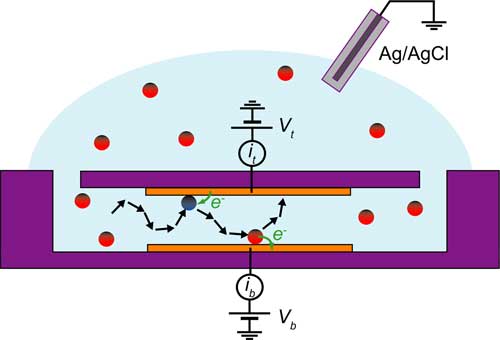
| Illustration of the device concept. Redox-active molecules undergo a random walk between two closely spaced electrodes, shuttling electrons from one electrode to the other. Vt and Vb are the potentials applied to the top and bottom electrodes with respect to a Ag/AgCl reference electrode immersed in the bulk reservoir, while it and ib are the corresponding measured currents. (Image: Dr. Lemay) | |
| In principle, the fabrication scheme for the device can be scaled down to the desired height needed to detect single molecules. Nobody has yet demonstrated such a device but the scientists are confident. | |
| "Now that the device works as designed, the next step is to scale it down so as to boost sensitivity further" says Lemay. "While not a conceptual problem, this does involve some work!" | |
| Zevenbergen points out that ultimately they would not only be able to detect single molecules in the device, but also discriminate between various species. | |
| "This is possible because the measured signal solely depends on the diffusion constant, which varies from species to species" he says. "A possible application would be that the device is integrated in a separation system in which single molecules of different species are detected and manipulated." | |
| "Our approach allows boosting the sensitivity of existing electrochemical biosensors based on reversible redox couples" adds Lemay. "In the longer term, we think that single-molecule sensitivity will also help to alleviate electrochemical sensors’ limited ability to discriminate between substances." | |
| The ability to confine, detect and manipulate individual electrons in solid-state devices was the basis for mesoscopic physics, which yielded many of the most exciting developments in solid-state physics over the last few decades. Achieving a similar level of control for ions is difficult because their natural length scale is the size of the ions themselves, about a nanometer. | |
| "Our work marks one of the first small steps toward this goal" says Lemay. "It’s new territory, nobody is sure yet what new physics there is to discover." | |
| By providing a clear route for the electrical detection of molecules in fluidic devices, the work of the Kavli Institute researchers paves the way for a range of experiments in fundamental statistical physics (eg. Maxwell demon devices in which individual ions are manipulated) and biophysics (eg. probing catalysis by single enzymes). Because the sensing device can readily be integrated into more complex nanofluidic circuits, it also provides a natural way to greatly enhance existing redox-cycling-based biosensors for lab-on-a-chip applications. | |
| These findings have been reported in the December 22, 2006 online edition of Nano Letters ("Mesoscopic Concentration Fluctuations in a Fluidic Nanocavity Detected by Redox Cycling"). | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
