| Posted: Jul 14, 2010 | |
Nanotechnology wound dressing automatically detects and treats infection |
|
| (Nanowerk Spotlight) In our previous Nanowerk Spotlight we reported on novel nanotechnology antimicrobial surface coatings and paints based on carbon nanotubes. While systems like these are highly effective against specific pathogens, they are in essence passive; i.e. they do not respond to their local environment and operate whether bacteria are present or not. | |
| Researchers in the UK have now conducted experiments that explored the elementary question of what it is that makes some bacteria pathogenic, and some not? Based on their findings, they have demonstrated that a simple vesicle (nanocapsule) system can be used as a 'nano-Trojan horse' for controlling bacterial growth and infection. Integrated into wound dressings, this novel material can automatically detect infection by pathogenic bacteria and respond to this by releasing an antibiotic into the wound, and changing color to alert medical staff. | |
| "The ultimate aim of our work is to attempt to engineer a 'smart' wound dressing system that only releases an encapsulated antimicrobial agent in the presence of pathogenic bacteria, without responding to harmless bacteria," >Toby Jenkins tells Nanowerk. "Basically, we have found a way so that we can use the property that makes some (not all) bacteria pathogenic by presenting them with capsules which bacterial secretion toxins attack (as if they are healthy tissue). Inside the capsules is an antimicrobial or a dye." | |
| In this work, reported in a recent issue of JACS ("A Thin Film Detection/Response System for Pathogenic Bacteria"),Jenkins and his colleagues from the Department of Chemistry at the University of Bath have shown that pathogenic bacteria can be used to be the agents of their own destruction by releasing toxins that rupture nanocapsules containing an antimicrobial agent. Although the skin is normally home to billions of 'friendly' bacteria, which are essential for a healthy skin, harmful bacteria pose a risk in case of injuries, which could lead to infections. This is a major problem with burn victims. | |
| "Our advanced wound dressing dressing will work by releasing antibiotics from nanocapsules triggered by the presence of disease-causing pathogenic bacteria, which will target treatment before the infection takes hold," Jenkins explains. "The dressing will also change color when the antibiotic is released, alerting healthcare professionals that there is infection in the wound. This is an important step in treating burns patients, particularly children, where infections can lead to toxic shock syndrome, a potentially fatal condition." | |
 |
|
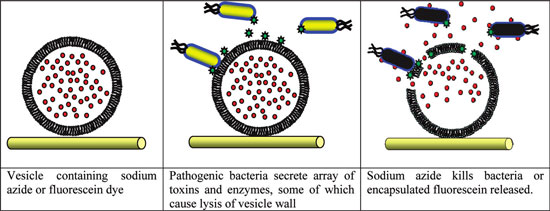
| Schematic of responsive antimicrobial system, showing an immobilized Giant Unilamellar Vesicle (GUV) with encapsulated antimicrobial. Toxins secreted by pathogenic bacteria lyse the vesicle and release the antimicrobial, concomitantly killing the pathogenic bacteria. (Reprinted with permission from American Chemical Society) | |
| The researchers have demonstrated the effectiveness of their system for two pathogenic species of bacteria, P. aeruginosa and S. aureus. A nonpathogenic strain of E. coli, which does not secrete toxins or membrane lysing agents, was used as a control system. | |
| The image above outlines the experimental approach explored in this work: Pathogenic bacteria secrete membrane damaging toxins/enzymes that lyse the lipid membrane of Giant Unilamellar Vesicles that are either attached to a surface or floating in aqueous suspension. | |
| Jenkins points out that the team pieced together what others have developed. Making vesicles and incorporating material inside is well know, as is the mode of action of various secretion toxins from pathogenic bacteria. Also, plasma treatment of nonwoven fabrics has been explored by a number of researchers and industries. Plasma provides a simple and inexpensive methodology for changing the hydrophilicity of a fabric. The University of Bath team linked these findings to come up with their advanced wound dressing technology. | |
| The problem with conventional dressings is that they have to be removed if the skin becomes infected in order to treat the infection. Not so with the, the nanocapsule dressing. It is automatically triggered to release antibiotics only when the wound becomes infected, meaning that the dressing will not need to be removed, thereby increasing the chances of the wound healing without scarring. | |
| In addition to a significant cost reduction, the proposed dressing will minimize the time required for the analysis of bacterial infection. This is particularly critical in burn wounds where delayed detection of bacterial infection can result in patient death. | |
| "The color change acts as an early warning system that infection is present, meaning we can treat it much faster, reducing the trauma to the child and cutting the time they have to spend in hospital" says Jenkins. | |
| The researchers hope to have a prototype dressing in 2-3 years, then work with engineers and companies to realize a commercial product. This will be done within the European Bacteriosafe consortium, | |
| Jenkins notes that a number of important problems regarding vesicle stability, tuning of response, manufacturability, and immunological response remain to be solved. It is unlikely, for example, that Giant Unilamellar Vesicles, which were used a nanocapsules for these sets of experiments, would be used in a final product, though part polymerized acetylene fatty acid/ phospholipid vesicles may find utility, this being the subject of current research. | |
| Watch a short video about the project: | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
