| Posted: Jul 15, 2010 | |
Using nanotechnology to unlock a fountain of bull |
|
| (Nanowerk Spotlight) Here is a perfect example of how someone, who apparently doesn't understand or care much about the science, writes a sensational press release hyping nanotechnology by cherry-picking information and distorting issues. And all that to sell a product that doesn't even have to do with nanotechnology. | |
| Two days ago we ran a press release from Thomson Reuters about a brief report they compiled on patent data relating to nanotechnology in the cosmetics industry. Now, Thomson Reuters is in the business of selling information and information services products and applications. Their press release basically is advertising for their IP Market Reports. There is nothing wrong with that. What is very wrong, though, is the nonsense and unbalanced take on certain aspects on nanotechnologies. Let's take a closer look. | |
| It already begins with the sensational title: Can Nanotech Unlock The Fountain of Youth?. That certainly catches the eye of the layperson. What exactly face creams, shampoos and sunscreens have to do with the "fountain of youth" remains unexplained. Oh, and they do make a reference to 'remote concepts' like nanorobotics. So let your imagination run wild! Little NanoStretchinators (trademark pending Nanowerk) that remove wrinkles from underneath the skin maybe? Or the fully automated Follicle-NanoSeeder that restores the shining body of the male scalp? | |
| The overview section of the pamphlet praises nanotechnologies in cosmetics more or less as the next big step in the development of humankind. One futurist guru gets quoted as saying: "Nanotech is one of the key design tools that will be used to create the largest industry in the 21st century: Health Enhancement. As humans yearn to improve themselves, they will be attracted to solutions in nanotech that will give them an edge." | |
| Not surprisingly, the report is somewhat coy when it comes to mentioning potential health and safety risks that are being raised by scientists. It even manages to make a reference to three government-funded activities to explore the risks of manufactured nanomaterials and give it a positive spin: "As commercial interests have increased in the category, so has investment into studying the health benefits of the technology." Note that it says health benefits – not risks. Here is more: | |
| "The U.S. Department of Health and Human Services earmarked a $15 million budget for the U.S. Food and Drug Administration (FDA) to develop new regulatory science to address the growth of nanotechnology in medical products and cosmetics. A separate initiative by the U.S. Environmental Protection Agency (EPA) has identified five nanomaterial types for further study that are widely used in products or have been recognized for their potential to be used in consumer applications. The European Commission has also begun studying the benefits of nanotechnology, hosting an international workshop on the use of nanotechnologies in cosmetics in the summer of 2009." | |
| Not a word about potential risks, or health and environmental concerns. But when you look at these three quoted studies you get a different message. The initiative by the EPA they are referring to actually "will determine whether these materials present a potential environmental hazard or exposure over their life cycles, and how these materials, when used in products, may be modified or managed to avoid or mitigate potential human health or ecological impacts." | |
| And while it sounds very positive that "European Commission has also begun studying the benefits of nanotechnology", the actual reference is to a workshop on regulatory issues regarding the use of nanomaterials in cosmetics. The specific objectives of this workshop were to | |
|
|
|
| Note that the Thomson Reuters guys talk about the benefits while what they are referring to mostly talks about safety, risk and studying the effects. That's quite a stretch to distort information to fit your message. | |
| Part of the report that actually contains some hard facts comes from data that was aggregated using the Thomson Reuters Derwent World Patents Index® (DWPISM) database, for patent research, and SAEGIS™, regarding trademarks, to identify global innovation and brand activity in nanotechnology for personal-care-products. Here is an example that shows the changes in patenting activity for cosmetics containing nanomaterials by product type from 2003 to 2009. | |
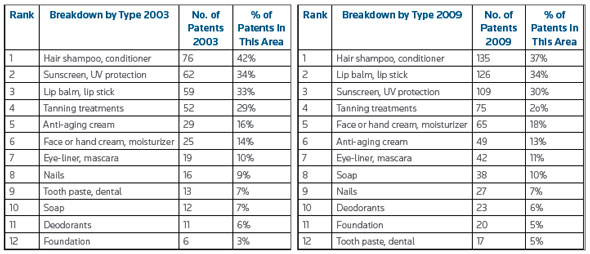 |
|
| Changes in patenting activity by product type from 2003 to 2009. (Source: Thomson Reuters, "Can Nanotech Unlock The Fountain of Youth?") | |
| Let's finish on an upbeat note. Here is a priceless quote from the report – no comment necessary: "Nanotechnology in personal-care items has escalated to new levels of sophistication. In the quest to preserve the appearance of youth, products as diverse as colored contact lenses, permanent makeup, and hair replacement therapy far outweigh living with the status quo." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
