| Posted: Aug 17, 2010 | |
Novel class of radially-aligned nanofibers promising for tissue regeneration |
|
| (Nanowerk Spotlight) Nanotechnology-enabled tissue engineering is a rapidly growing field. At the core of tissue engineering is the construction of scaffolds out of biomaterials to provide mechanical support and guide cell growth into new tissues or organs. In particular, electrospun biodegradable polymeric nanofibers are being used in scaffolds for engineering various tissues such as nerves, cartilages or bone (read more: "One day doctors will grow new bones with nanotechnology"). Electrospinning is a fabrication technique which can produce nanoscale fibers from more than 100 different polymers. The electrospun nanofibers are typically collected as nonwoven mats with random orientation. | |
| A new study by researchers at Washington University in St. Louis has now demonstrated the fabrication of a novel class of nanofiber scaffold composed of radially-aligned, electrospun nanofibers and also demonstrated the unique application of these materials as effective biomedical patches/scaffolds that could prove to be beneficial during neurosurgery. | |
| "Such a scaffold represents a significant departure from existing scaffolds fabricated through traditional electrospinning techniques," Younan Xia, the the James M. McKelvey Professor for Advanced Materials in the Department of Biomedical Engineering, tells Nanowerk. "Specifically, individual nanofibers within these novel scaffolds are not limited to simple orthogonal patterns imparted by uniaxial-alignment, but instead are capable of achieving any number of organized radial orientations around a central point electrode." | |
| Essentially, the novel method of electrospinning described in a paper in the August 9, 2010 online issue of ACS Nano ("Radially Aligned, Electrospun Nanofibers as Dural Substitutes for Wound Closure and Tissue Regeneration Applications") extends nanofiber patterning techniques beyond simple Cartesian patterns, and enables the creation of advanced radial patterns of organized, aligned nanofibers. | |
 |
|
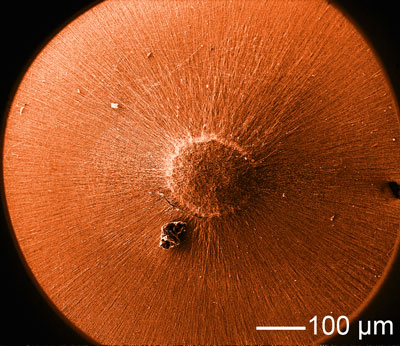
| SEM image of a radially aligned, electrospun nanofiber scaffold. (Image: Xia Lab, Washington University in St. Louis) | |
| "Additionally," says Jingwei Xie, first author of the ACS Nano paper, "our study demonstrates that radially-aligned nanofibers provide an optimal substrate capable of directing and enhancing wound healing and tissue regeneration. Radial-alignment of individual nanofibers within these novel scaffolds offers a unique method of presenting organized topographical cues useful in directing cellular migration from the periphery of native tissue directly to the center of the nanofiber patch." | |
| As a result, it has been hypothesized that radially-aligned nanofiber scaffolds would increase the speed of cellular ingrowth, and as a result support faster wound healing and recovery. In their work, the Washington University team confirms this hypothesis by applying these novel scaffolds within ex vivo duraplasty models (dura is the outermost of the three layers of the meninges surrounding the brain and spinal cord and is responsible for keeping in the cerebrospinal fluid). | |
| The researchers showed that patches composed of radially-aligned nanofibers promoted faster ingrowth of dural fibroblasts into induced dural defects than either patches composed of randomly oriented nanofibers or random collagen matrices. | |
| Overall, this new study suggests that this novel class of radially-aligned nanofiber scaffolds represents a unique advancement in the design of biomedical patches useful in repairing wounds and defects in many types of tissue throughout the human body. | |
| "Ideally, further investigation into these scaffolds may lead to the creation of nano-patterned scaffolds, dressings, and patches capable of modulating cellular migration and function as a means of enhancing wound healing and improving patient outcomes" says Xia. | |
| Xie recounts how this present study was motivated by a recent collaboration established between the Department of Biomedical Engineering at Washington University and the Department of Neurosurgery at Washington University School of Medicine: "In a meeting between Matthew R. MacEwan, Dr. Ray, Dr. Xia, and myself an idea was proposed to produce a more effective biomaterial patch for use in repairing large-scale dural defects. Specifically, we were interested in producing a scaffold that not only possessed superior handling properties but additionally enhanced wound healing and tissue regeneration. While electrospun nanofiber scaffolds represented an obvious choice, due to their compliance and ease of handling, it was unclear how nanofibers could be utilized to enhance cellular ingrowth into a tissue defect." | |
| The scientists knew that none of the uniaxially-aligned or randomly-oriented nanofiber scaffolds fabricated previously was able to recapitulate the radial cell migration known to occur from the periphery of a tissue defect to the center of the defect. | |
| "By experimenting with various novel collector systems, we eventually created a scaffold composed purely of radially-aligned nanofibers oriented around a point electrode placed within the center of the scaffold" explains Xie. "These novel radially-aligned scaffolds provided the ideal material for use as a biomedical patch in that they were both easy to handle, and utilized the organized nano-topography of radially aligned nanofibers to guide migrating cells in a physiologically-relevant manner from the periphery of the defect into the body of the scaffold." | |
| While this current study specifically demonstrates the potential of these scaffolds to serve as effective dural substitutes, superceding even gold-standard dural matrices, this technology can be broadly applied in a number of biomedical applications. | |
| According to the researchers, radially-aligned nanofiber scaffolds could provide an ideal substrate for effective biomedical patches, dressing, scaffolds, and implants capable of enhancing wound healing in any number of human tissues. They may be of particular use in repairing defects in a number of laminar tissues, including the skin, bowel, and connective tissues. | |
| Overall, from a perspective of nanotechnology in medical applications, the results of this study demonstrate that nanofabricated materials possess significant potential as effective biomedical devices, and that nano-patterned materials and surfaces can modulate cellular activity even in more complex physiological micro-environments. | |
| "Nanofabricated and nanopatterned materials are at the forefront of biomaterial research" says Xia. "The ability of these materials to modulate and control cellular activity by replicating sub-cellular features of the extracellular environment offers tremendous potential. Yet, despite the capability of this unique class of materials, very few nanofabricated materials have been successfully implemented in clinical scenarios. We believe that the next phase of research in the field of nanofabricated biomaterials will focus primarily on studying the function of these materials in more complex physiological settings, and simultaneously translating many of these advances in nano-fabrication and nanotechnology into effective medical devices and therapeutics capable of improving clinical care and patient outcomes. The specific challenges could be the translation from in vitro study to in vivo study or from in vivo study to clinical study as the scenario is more complex." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
