| Posted: Oct 07, 2010 | |
Nanomechanical sensor can detect cholera |
|
| (Nanowerk Spotlight) One of the tools developed by molecular biologists to study cell membranes and their associated proteins is a synthetic membrane model called Nanodisc. This is a self-assembled phospholipid bilayer disc of about 10 nm diameter, wrapped in a membrane scaffold protein belt. Nanodiscs render amphipathic and hydrophobic molecules easily soluble, offering transformative innovations across a broad range of applications in both in vivo delivery of therapeutics, diagnostic and imaging agents as well as for in vitro drug discovery. They have become important tools in analyzing membrane proteins, which are the most important target for present-day drug discovery programs. | |
| In contrast to the liposomes traditionally used for solubilizing membrane associated macromolecules – which form large complexes which are heterogeneous, unstable and can skew the phenomena observed on the surface – nanodiscs provide homogeneous discs with controlled stoichiometry of the incorporated protein. | |
| Further developing the nanodisc toolbox, researchers have now demonstrated the efficacy of Nanodiscs receptors for the nanomechanical detection of cholera. | |
| "Our contribution promises new opportunities in evaluating the binding of proteins 'directly' to membrane protein receptors stabilized in Nanodiscs, not to their antibodies," Soo-Hyun Tark, a Postdoctoral Fellow in the Department of Materials Science and Engineering at Northwestern University, tells Nanowerk. "Although antibodies have shown to provide high sensitivity and selectivity for the target analytes against which they are specifically generated, it may not be always possible or efficient to generate antibodies against all the proteins that naturally bind to membrane protein receptors." | |
| He points out that, in contrast, Nanodiscs can be considered as a universal platform to directly incorporate, solubilize and immobilize the membrane protein receptors for investigating the protein and membrane receptor interactions. | |
| "When combined with a signal transduction approach such as microcantilevers providing label-free and real-time measurements, we believe many applications can be found where monitoring these binding events is critical" says Tark. | |
| The Northwestern team, led by Stephen Sligar and Vinayak Dravid, reported their findings in the October 4, 2010, online issue of Nanotechnology ("Nanomechanical detection of cholera toxin using microcantilevers functionalized with ganglioside nanodiscs"). | |
 |
|
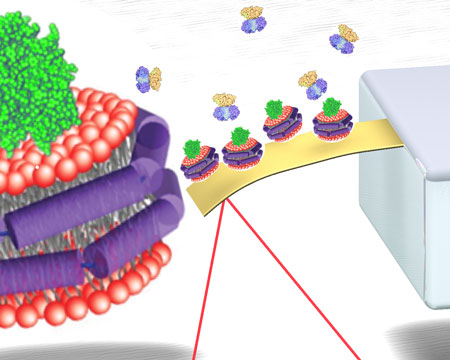
| Nanomechanical detection platform functionalized with receptor-incorporated Nanodiscs. (Image: Soo-Hyun Tark and Aditi Das) | |
| Nanomechanical sensors based on microcantilevers have emerged and gained increasing attention as a sensitive and label-free detection platform for a wide range of chemical and biological interactions. These microcantilever look a bit like tiny springboards (about 500 µm long and sometimes less than 1 µm thick) and they bend in response to different forces. Coating one side of such a microcantilever with a layer of high affinity receptors can transduce the binding of target molecules into a nanomechanical response of the cantilever (basically making the 'springboard' vibrate, albeit only by hundreds of nanometers). | |
| When the cantilever signal transduction platform operates in the static deflection mode, the cantilever's response to such a binding event is deflection in the range of a few to hundreds of nanometers as a result of the adsorption-induced surface stress and consequently the development of a bending moment. This cantilever deflection can then be measured either optically (using lasers) or electrically (by using piezoresistive or MOSFET-embedded microcantilevers). | |
| Although the microcantilever detection platform provides sensitive transduction of specific molecular binding into the nanomechanical cantilever response in real-time, creating a robust and efficient receptor layer on the cantilever surface has remained a challenge. | |
| "The key finding from our work is that the microcantilever-based nanomechanical sensing platform operating in the static deflection mode can be synergistically combined with Nanodiscs to directly measure the binding events between proteins and membrane associated receptors incorporated into the Nanodiscs" explains Tark, who is first author of the above-mentioned paper. "The Nanodiscs are employed to directly incorporate, solubilize and immobilize the membrane protein receptors. Such an integrated system enables sensitive transduction of the specific molecular binding events into the nanomechanical cantilever response in real-time and in a label-free fashion." | |
| Nanodiscs allow immobilization of the membrane-associated protein receptors on the cantilever surface while maintaining their stability and bioactivity otherwise difficult to achieve using currently available methods. Thus, integrating receptor-incorporated Nanodiscs to microcantilever could offer new avenues to create a stable receptor layer of important membrane proteins for a functioning sensor platform. | |
| To validate and evaluate the efficacy of the nanodisc-microcantilever approach, the researchers investigated the detection of cholera toxin as a model system using ganglioside GM1-incorporated nanodiscs immobilized on the static mode microcantilevers. | |
| "We show that the label-free detection of cholera toxin is possible by measuring the stress-induced deflection of the cantilever, which confirms the potential of the system combining the advantages of Nanodiscs and the nanomechanical sensor platform for applications in detecting toxins and investigating the association between macromolecules and membrane proteins in Nanodiscs" says Tark. | |
| The approach opens up new opportunities in investigating the biomolecular interactions particularly for those involving the membrane associated receptors that are very important drug targets. | |
| "When combined with the unique multiplexing capability of the microcantilever platform, we envision such a Nanodisc-based detection approach may find many significant applications in drug discovery and pathogen monitoring" says Tark. | |
| The team is now working on improving the sensitivity of their nanomechanical detection platform by optimizing the cantilever design and material as well as by implementing enhanced signal readout mechanisms such as integrated piezoresistors and transistors. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
