| Posted: Jun 07, 2011 | |
A 'green' nanomaterial to remove organic impurities from waste water |
|
| (Nanowerk Spotlight) Water treatment is important for human consumption and environmental protection. Non-trivial purification of water involves removal of toxic ions, organic impurities, microbes and their by-products as well as scooping oil spills. The removal of organic contaminants from water is a major industrial concern. The contaminated water from industries usually contains considerable amounts of organic hydrocarbons such as BTEX (benzene toluene, ethyl benzene and xylene) prohibiting direct release into natural water bodies. The dissolved organic compounds cause bacterial growth, odor generation and biofouling bringing limitation on reuse (see: "The Challenge of Micropollutants in Aquatic Systems"). | |
| The challenging goal here is to detect, decompose and remove contaminants present usually in low concentrations (see: "Science and technology for water purification in the coming decades"). Towards this end, different types of sorbent materials have been developed to date, the most common being activated carbon. Though the use of activated carbon is still considered to be one of the best method, the disposal of adsorbed contaminants along with the adsorbent is a major concern. Thus, the decomposition of the adsorbed organic contaminants and regeneration of the adsorbent is a critical step to a cost-effective use of the porous adsorbents. | |
 |
|
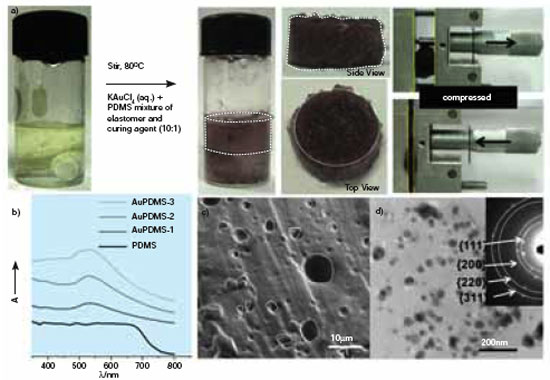
| Fig.1: Green Synthesis of AuPDMS nanocomposite: (a) A highly compressible AuPDMS foam (b) The UV-visible spectra of the foam exhibit the characteristic peak at 534 nm (the increasing intensity in the order of nanoparticle loading AuPDMS-1 to AuPDMS-3) (c) The SEM image of AuPDMS foam with irregular and interconnected pores of size (2-20 µm) distributed all over the surface. (d) TEM image and ED pattern shows crystalline Au nanoparticles of 10-30 nm size. (Reprinted with permission from Wiley-VCH Verlag GmbH & Co. KGaA, R. Gupta et al., ChemSusChem, 4, (2011), DOI: 10.1002/cssc.201000410) | |
| We have recently come up with an innovative method for organic pollutant removal from waste water (see: "Removal of Organic Compounds from Water by Using a Gold Nanoparticle–Poly(dimethylsiloxane) Nanocomposite Foam"). The issue of water purification was dealt from an environmental perspective, using a Au nanoparticle-polydimethylsiloxane (AuPDMS) nanocomposite in the form of a foam. With this nanocomposite, one is able to bring together the unique properties of Au nanoparticles and PDMS elastomer. | |
| The synthesis is a single step room temperature process involving reduction of Au3+ ions from KAuCl4 precursor in PDMS matrix in presence of water, thus eliminating the need of external reducing and capping agents. It is essentially a green process (Fig. 1). The Au nanoparticle loading in the PDMS foam has been optimized (0.03 wt%) to attain a low density (~1.1 g/cm3), high compressiblity (~105%) as well as pore volume (~5.45 cm3/g), towards making it a high performance material. | |
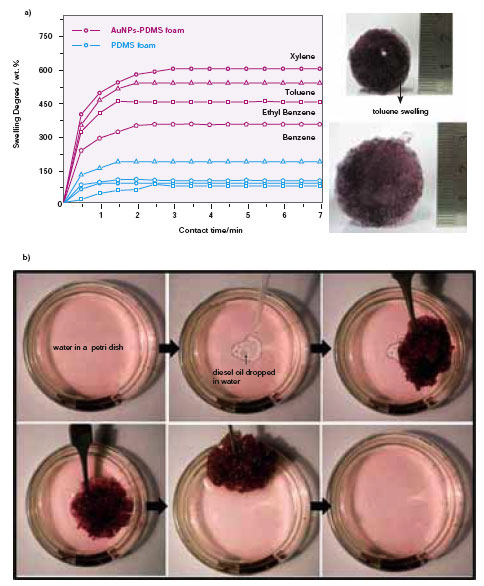
| The performance of AuPDMS nanocomposite has been tested against BTEX for comparison, where the sorbent capacity is found to be nearly six times higher compared to PDMS foam without Au nanoparticles (Fig.2a). The material is ideally suited for removal of organics and oil spills (Fig. 2) because of its high and fast swelling capacity with longer retentivity, as demonstrated for diesel oil on water. | |
 |
|
| Fig. 2: Removal of industrial organic pollutants (BTEX) and oil spills from water: (a) uptake curve for benzene, toluene, ethylbenzene and xylene present in contaminated water for PDMS and AuPDMS foams. (b) Demonstration for removal of diesel oil in water. Diesel uptake capacity of AuPDMS is 125% of its weight and the uptake happens within minutes. (Reprinted with permission from Wiley-VCH Verlag GmbH & Co. KGaA, R. Gupta et al., ChemSusChem, 4, (2011), DOI: 10.1002/cssc.201000410) | |
| The active role of Au nanoparticle surface for sulfur bonding has been exploited for effective removal of odorous sulfur containing molecules (Fig. 3). The AuPDMS foam has also been utilized for highly selective removal of an organic sulphide (thioanisole) from water with consistently good activity which is rarely found in literature (see: "Odor Control in Wastewater Treatment: The Removal of Thioanisole from WaterA Model Case Study by Pulse Radiolysis and Electron Beam Treatment"). The extraction of thioanisole from contaminated water was performed using AuPDMS foam by treating thioanisole contaminated water with the foam for set intervals of time. Between the intervals, the foam was heated for small interval in air at 300°C to remove the adsorbed thioanisole and regenerate the foam. | |
| The concentration versus contact time plot followed the first order kinetics with rate constant estimated to be 0.15 hr-1. The reusability of AuPDMS foam was further tested by conducting several cycles of thioanisole removal. We have also addressed the efficiency and recoverability of material. Additionally, the AuPDMS foam can be recycled and reused repeatedly for many cycles by heating in air at 300°C. | |
 |
|
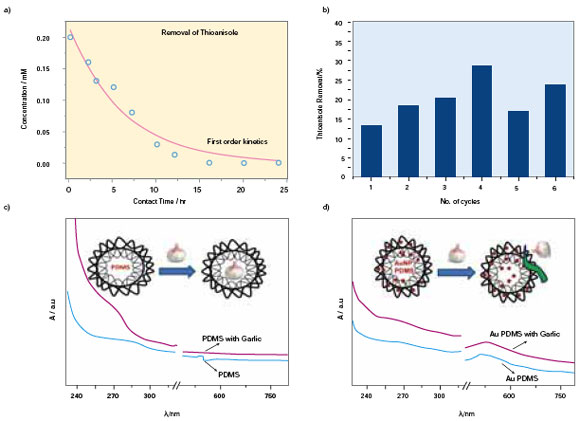
| Fig. 3: Removal of odorous molecules from contaminated water and odor scavenging from garlic extract: (a) Change in concentration of thioanisole on treatment with AuPDMS foam (b) different cycles of thioanisole removal by AuPDMS foam. Absorption spectra of (c) adsorbed garlic in PDMS foam exhibiting a broad feature around 258 nm (d) AuPDMS foam after garlic treatment shows no change. (Reprinted with permission from Wiley-VCH Verlag GmbH & Co. KGaA, R. Gupta et al., ChemSusChem, 4, (2011), DOI: 10.1002/cssc.201000410) | |
| Its ability was also tested in eliminating odor from water that contained natural garlic extract as seen in Fig. 3c and 3d. Moreover,the garlic solution got deodorized almost completely on treatment with AuPDMS, which could be sensory analysed. Interestingly, with the plain PDMS foam (control), the malodor continued to emerge even after a month! The AuPDMS foam was observed to be acting as a odor scavenging system and may therefore be of interest for food packaging. The Au nanoparticle PDMS composite is indeed an attractive material for water based pollutants and odorous molecules. It can be moulded into any shape during or after synthesis and fitted into water channels or installed in water bodies for practical applications. | |
| By Ritu Gupta and Giridhar U. Kulkarni, Chemistry and Physics of Materials Unit and DST Unit on Nanoscience, Jawaharlal Nehru Centre for Advanced Scientific Research, Jakkur, Bangalore 560064, India. Email: [email protected] & [email protected] . | |
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
