| Posted: Oct 15, 2012 | |
Remotely activating biological materials with nanocomposites |
|
| (Nanowerk Spotlight) The heating properties of iron oxide nanoparticles have been exploited through the years for use in cancer therapy, gene regulation, and temperature responsive valves. These applications have demonstrated the versatility of iron oxide nanoparticles, but they had rarely, if ever, been used to enhance the activity of thermophilic enzymes. | |
| Thermophilic enzymes are highly stable biomolecular systems that are excellent tools due to their thermostability and long-term activity for extended lifetime uses in the field and other applications. | |
| Traditionally, these enzymes are heated in an oven or water bath at a specified temperature. These heating methods are not efficient and much of the heat can be lost due to diffusion limitations of the heat to the sample. | |
| New work by researchers in the U.S. addresses the problem of remotely activating biological materials with a higher efficiency than conventional methods such as water baths or convection ovens. | |
| "In our work, we fabricated a hydrogel in which we chemically immobilized a thermophilic dehalogenase and encapsulated iron oxide (Fe3O4) nanoparticles," Sylvia Daunert, Professor and Lucille P. Markey Chair, Department of Biochemistry and Molecular Biology at the University of Miami, explains to Nanowerk. "Previous studies have shown that these nanoparticles heat up when in the presence of an alternating magnetic field (AMF). We used the hydrogel nanocomposites in the AMF to find the field strength for optimum enzymatic activity. Additionally, we heated the hydrogel nanocomposites in a water bath at the optimal temperature for enzymatic activity." | |
| Comparing these results, it was found that the enzyme was two times more efficient when heating in the AMF as compared to heating in a water bath in one-third of the time. | |
| Daunert and her team reported their findings in the September 18, 2012 online edition of ACS Nano ("Nanoparticle-Mediated Remote Control of Enzymatic Activity"). | |
| Daunert points out that this heating method is localized as compared to other heating methods. "Furthermore, by using an AMF, which is not limited by penetration depth for uses in vivo like other nanomaterials, we envision that this method could be expanded for personalized therapeutics, bioremediation, catalysis, filtering devices, separations, etc. In short, this application could be potentially applied to any system where you need to turn on and off bioactivity." | |
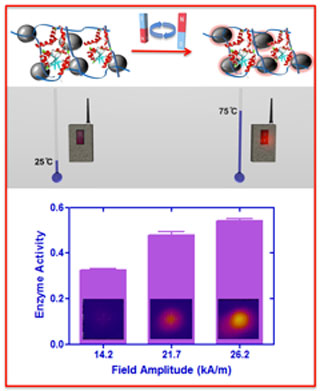 |
|
| Top: Thermophilic enzymes can be encapsulated into a hydrogel network with iron oxides nanoparticles. Remote-controlled actuation is achievable by exposing the materials to an alternating magnetic field. Bottom: The activity of the enzyme is tunable dependent upon the field amplitude. (Figure: Leslie D. Knecht, University of Miami) | |
| This work was the result of collaboration between Daunert's lab and J. Zach Hilt's group in the Department of Chemical and Materials Engineering at the University of Kentucky. | |
| "Dr. Hilt's group used an AMF to heat the Fe3O4 nanoparticles for hyperthermia treatment in cancer patients" says Daunert. "My lab focused on recombinant proteins for biomedical and environmental analysis. The enzyme had recently been characterized in my lab and we felt it was an ideal enzyme to show the proof of concept that we could efficiently heat and activate an enzyme remotely using Fe3O4 nanoparticles." | |
| Previous research had already shown that by combining the remote heating capabilities of magnetic nanoparticles with the stimuli responsive properties of hydrogels, multifunctional materials can be designed for targeted applications such as drug delivery or hyperthermia cancer treatment (see for instance our previous Nanowerk Spotlight: "Remote-controlled nanocomposite for on-demand drug delivery inside the body"). | |
| What is new in this work is that the responsive hydrogel nanocomposite system includes a thermophilic enzyme that can be remotely switched on or off. In their paper, the team focuses on using a thermophilic dehalogenase for remote controlled dehalogenation of samples. By encapsulating the enzyme into the hydrogel, it allows for the use of the biocatalyst through many enzymatic cycles with different substrates without the need for complicated separation and/or regeneration steps. | |
| "Although this current work focuses on bioremediation, it can be expanded to other applications where thermophilic proteins are employed," says Daunert. "Additionally, the enzyme and nanoparticles in our work were immobilized in a hydrogel network but for future applications, various matrices can be used such as weaving the protein into textiles or incorporation into thin films." | |
| This means that, due to the numerous available methods and chemistries used to functionalize proteins, there are many materials that can be used as platforms to incorporate both the nanoparticles and protein of interest for a variety of applications. | |
| For instance, the scientists envision that their technique could be applied to molecular biology techniques such as using thermostable DNA polymerase for polymerase chain reaction. Remote heating may be plausible in industrial settings where thermophilic enzymes are used for processes such as hydrolysis of starches, cleavage of proteins, or use of thermophilic lipases. The benefit of using a localized heating method allows for less energy input for a greater catalytic output, which could provide cost-savings for some of these applications. | |
| Daunert notes that the library of thermophilic organisms that are being discovered is growing. "As this number grows, the breadth of capabilities of this system can be extensive, limited only by the number and type of thermophilic proteins available. Incorporation of thermophilic enzymes into additional platforms beyond hydrogels represents an important advance for designer applications where additional experimental challenges will be met and further research will be required." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
