| Posted: Jul 02, 2013 | |
Preserving the edge magnetism of graphene nanoribbons |
|
| (Nanowerk Spotlight) Graphene nanoribbons are strips of graphene that are only a few nanometers wide. Graphene has two distinct types of edges produced when it is cut – armchair type or zigzag type – which correspond to the two crystal axes of graphene. In contrast to sheet graphene which is actually semimetal, both zigzag and armchair nanoribbons have a nonzero band gap like those in semiconductors, which has been confirmed both theoretically and experimentally. In addition to distinct electronic properties, and depending on the width and the angle at which they are cut, graphene nanoribbons also have distinct chemical, magnetic, and optical properties. | |
| Edge magnetism is a unique property of graphene ribbons that has been predicted by theory, but not yet directly confirmed experimentally. This could be due to the instability of the ideal zigzag graphene nanoribbons (zGNRs) because, to preserve the edge magnetism, the edge sites of zGNRs should have the pure sp2 coordination. Generally, hydrogen is chosen as terminal group for zGNRs to saturate the dangling bonds. However, a theoretical study revealed that pure sp2 coordinated edges of hydrogen terminated zGNRs can be stabilized only at extremely low hydrogen concentration, which is rather challenging experimentally. | |
| If researchers want to utilize zGNRs in spintronics, they first need to figure out a suitable termination group for zGNRs. The often used hydrogen atom termination is not a good choice since hydrogen terminated zGNRs can only be stabilized at extremely low hydrogen concentrations. | |
| In new work, researchers from the Center for Advanced Nanoscale Materials in the Department of Chemistry at the University of Puerto Rico and Institute of New Energy Material Chemistry, the Key Laboratory of Advanced Energy Materials Chemistry (Ministry of Education) at Nankai University, designed special boundaries for zGNRs that could become both stable and maintain the edge magnetism. | |
| "We demonstrated theoretically that ethylene (C2H4), a very simple and common organic molecule, is the ideal terminal group for zGNRs in terms of preserving the edge magnetism," Zhongfang Chen, an Associate Professor in the Department of Chemistry at the University of Puerto Rico, tells Nanowerk. "By computation from density functional theory, we confirmed our hypothesis and explained it by Clar's rule. This is a beautiful demonstration of a well-thought-out computational experiment." | |
| In a paper published in the June 19, 2013, online edition of Scientific Reports ("Preserving the Edge Magnetism of Zigzag Graphene Nanoribbons by Ethylene Termination: Insight by Clar's Rule"), Chen and his team used density functional theory to systemically study the energetics and electronic properties of C2H4-zGNRs with different edge configurations with the goal of preserving edge ferromagnetism. | |
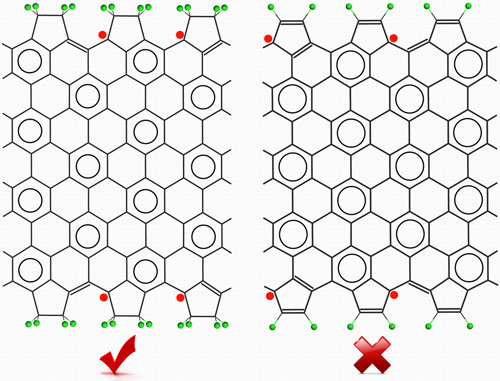 |
|
| C2H4 terminating groups can well preserve the edge magnetism of zGNRs, while C2H2 termination with pure sp2 edges can not. (Courtesy Dr. Yafei Li) | |
| "The C2H4 terminated zGNRs (C2H4-zGNRs) with pure sp2 coordinated edges can be stabilized at rather mild experimental conditions, and meanwhile reproduce the electronic and magnetic properties of those hydrogen terminated zGNRs," explains Chen. "The electronic structures and relative stability of C2H4-zGNRs with different edge configurations can be well interpreted by employing the Clar?s rule. Compared with hydrogen terminated zGNRs, the enhanced stability of sp2 coordinated edges of C2H4-zGNRs is attributed to the multiple edge hyperconjugation interactions. Moreover, taking C2H2 termination as an example, we demonstrated that even pure sp2 termination is not a guarantee for edge magnetism if the terminal group can couple to the π-electron system of zGNRs." | |
| Experimentally, this work will pave the way for the applications of zGNRs in spintronics; theoretically, it should encourage the use of Clar?s rule to describe the electronic structures of graphene materials. | |
| "If we want to utilize zGNRs in spintronics, we need develop new methods to produce zGNRs with smoother edges," concludes Chen. "And the realization of C2H4-terminated zGNRs is much more feasible, though still experimentally challenging." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
