| Posted: Mar 16, 2006 | |
Understanding the role of carbon nanotubes as water carriers |
|
| (Nanowerk News) "A key step for developing novel nanomedicine technologies consists in delivering small amounts of aqueous solutions through the hollow interior of carbon nanotubes" Professor Alberto Striolo explains to Nanowerk. "The properties of water, structure and dynamics, for example, are expected to change dramatically when bulk water is confined within nanotubes whose diameter is of the same order of magnitude as the size of one water molecule" he adds. | |
| Striolo, a professor at the School of Chemical, Biological, and Materials Engineering at the University of Oklahoma, has conducted research that documents some of these changes, focusing on the diffusion properties of confined water: e.g. how fast does water move within a carbon nanotube (CNT)? His findings, titled "The Mechanism of Water Diffusion in Narrow Carbon Nanotubes" were published in the March 2, 2006 online edition of Nano Letters. | |
| Research like Striolo's is crucial for achieving the complete understanding of the properties of confined water that is undoubtedly necessary for the design of novel nano-machinery such as nano-syringes or CNT-embedded membranes for the controlled delivery of nanometer quantities of aqueous solutions. | |
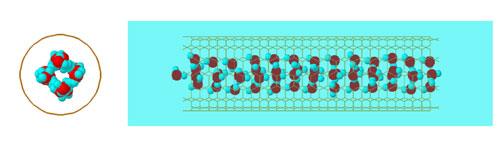 | |
| Representative simulation snapshots for water confined in (8:8) SWNTs. Shown is the view parallel (left) and that perpendicular (right) to the pore axis. The carbon atoms are not shown for clarity. The yellow lines connect the centers of nearest-neighbor carbon atoms. Oxygen atoms are shown in red, hydrogen atoms are shown in light blue. (Source: University of Oklahoma, Alberto Striolo) | |
| Scientific interest was centered initially on water's behavior within short, narrow CNTs. It was found that water molecules move occasionally along the nanotube axis via bursts of hydrogen-bonded clusters of molecules. Experimental observations of water diffusion through bacterial potassium channels seem to corroborate the theoretical predictions. | |
| In studying arrays of aligned CNTs extremely high flow velocities have been experimentally observed when the CNTs are of approximately 7 nm in diameter. These studies find theoretical corroboration in earlier molecular simulations for the osmotic water transport through carbon nanotube membranes, in which the fast diffusion of water through the CNTs was ascribed to the single-file diffusion mechanism. | |
| The goal of Striolo's studies are to understand the mechanism of water diffusion through infinitely-long narrow CNTs and to understand whether or not the transport properties of confined water differ from those of simple fluids. | |
| One conclusion of the simulations suggest that the molecular mechanism responsible for water diffusion does not depend substantially on the density of confined water molecules, but that the organization of water molecules within a cluster (whether ordered or not) does influence the mechanism of motion. | |
| Striolo used a software package to conduct molecular dynamics simulations in order to understand if and when single-file diffusion can occur for confined water molecules. He demonstrated that the transport of water molecules through an infinitely-long narrow CNT occurs via a coordinated motion, independently on the degree of filling of the nanotubes. | |
| The ballistic motion reported in Striolo's paper lasts for exceptionally long times (up to 500 ps) and it is due to a molecular mechanism that results from long-lasting hydrogen bonds between adsorbed water molecules, weak carbon-water attractive interactions, and smooth SWNTs surfaces. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
