| Posted: Dec 11, 2007 | |
First nanotechnology genotoxicity tests find that carbon nanotubes could damage DNA |
|
| (Nanowerk Spotlight) Carbon nanotubes (CNTs) have shown promise as an important new class of multifunctional building blocks and innovative tools in a large variety of nanotechnology applications, ranging from nanocomposite materials through nanoelectronics to biomedical devices (e.g. gene and drug carriers). The recent rapid development in nanotechnology has renewed the pressing demand for large-scale production of CNTs for potential applications in commercial products. | |
| The number of industrial-scale facilities for the increasingly low-cost production of multi-walled carbon nanotubes (MWCNTs) continues to grow, and with that the professional and public exposure to MWCNTs is expected to increase significantly in the coming years. Owing to their unusual one-dimensional hollow nanostructure and unique physicochemical properties, CNTs are particularly useful as novel drug delivery tools and imaging agents. However, such biomedical, and many other related, applications will not be realized if there is no proper assessment of the potential hazards of CNTs to humans and other biological systems. | |
| This situation prompted a group of researchers to carry out the first genotoxicity study of nanomaterials. Although the health effects of nanomaterials have attracted considerable attention, the scientific community has thus far focused primarily on the studies of nanomaterials toxicity at the cellular level. Very little is known about the toxicity at the molecular level, or genotoxicity, of nanomaterials in mammalian cells. | |
| Researchers at the University of Dayton have assessed the DNA damage response to MWCNTs in mouse embryonic stem cells (ES). This new work emphasizes the importance of careful scrutiny of the genotoxicity of nanomaterials. | |
| "Our results suggest that careful scrutiny of the genotoxicity of nanomaterials is needed even for those materials, like multi-walled carbon nanotubes, that have been previously demonstrated to have limited or no toxicity at the cellular level" Dr. Liming Dai tells Nanowerk. "The results of our study also provide strong support for the use of p53 as a biomarker for preliminary screening of genotoxicity of nanomaterials." | |
| Dai explains that, in response to DNA damage, eukaryotic cells, including ES cells, have developed several mechanisms to protect genomic integrity. In the presence of damaged DNA for instance, the p53 protein is activated by protein phosphorylation as a master guardian that activates cell cycle checkpoints and triggers cell cycle arrest to provide time for the DNA damage to be repaired. | |
| "Enhanced expression of p53 may also trigger cell death by apoptosis if the DNA damage is beyond repair, while under normal conditions (absence of DNA damage) p53 is expressed at low levels" he says. "The close relationship between p53 activation and DNA damage makes p53 the molecular marker of choice for assessing the genotoxicity of MWCNTs to mouse ES cells." | |
 |
|
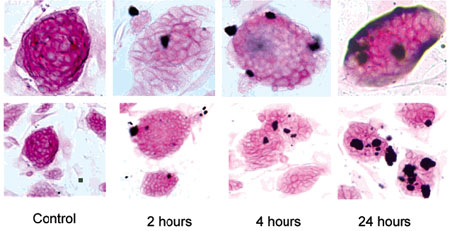
| The images of MWCNT uptake and induce differentiation and apoptosis in mouse ES cells following MWNT treatment. Mouse ES cells were treated with 100 µg/mL of MWCNTs for 2, 4, and 24 hours. ES cells (control and exposed) were washed with 1X PBS and fixed with 4% paraformaldehyde. AP stain results showed that some ES cells began to lose AP turning to white in color compared with the untreated cells, indicating that the MWCNT reduced the stem cell marker expression. The images were captured by inverted microscope at 10x (bottom row) and 20x (top row) magnification. (Reprinted with permission from American Chemical Society) | |
| The collaborative work between the University of Dayton research groups of Dai, Wright Brothers Institute Endowed Chair in Nanomaterial, and Dr. Yiling Hong, an Assistant Professor in the Department of Biology, has demonstrated for the first time that MWCNTs can accumulate in mouse ES cells, and that MWCNTs lead to the generation of free radicals called reactive oxygen species (ROS). Free radicals can chemically alter DNA bases and cause DNA damage within the cell. | |
| As the researchers report in their paper in the November 29, 2007 online edition of Nano Letters ("DNA Damage Induced by Multi-walled Carbon Nanotubes in Mouse Embryonic Stem Cells"), the DNA damage induced by MWCNTs was indicated by Western blot analysis of 8-oxoguanine-DNA glycosylase 1 (OGG1) and by the induction of two key double-strand break repair proteins, Rad 51 and XRCC4. Furthermore, the MWCNT treatment was found to cause a two-fold increase in the mutation frequency in mouse ES cells. | |
| "Our work represents the first genotoxicity study of nanomaterials and indicates the need for careful scrutiny of the toxicity of nanomaterials at the molecular level" says Dai. "Because of its novelty and potential implication to other nanomaterials, we feel that this work will spur unforeseen ideas and investigations by other groups worldwide to establish the field of nanomaterials genotoxicity." | |
| The methodology developed in Dai and Hong's study is highly versatile for testing genotoxicity of many other nanomaterials. With the large number of nanomaterials already developed, and more to be synthesized, this study opens up avenues for genotoxicity studies of nanomaterials for various potential applications. | |
| Recent studies suggest that the biocompatibility of nanomaterials depends strongly, among many other factors, on surface functional groups. Although controlled functionalization of nanomaterials is still a challenge, Dai says that the cytotoxicity/genotoxicity of nanomaterials could be minimized by functionalization them with biocompatible surface moieties. "Overcoming this challenge should lead to a number of practical applications of nanomaterials, including carbon nanotubes, even in biomedical systems." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
