| Posted: May 20, 2008 | |
Fingerprinting nanoparticles to assess cytotoxicity |
|
| (Nanowerk Spotlight) High content analysis (HCA) is a powerful platform that combines cell-based assays with traditional microscopy and automated, sophisticated image processing and analysis software. This technology is capable of using living and fixed cells, typically with fluorescently labeled antibodies and reagents. It has been widely adopted in the pharmaceutical and biotechnology industries for target identification and validation. HCA has made particular inroads into R&D applications where high throughput screening has proven inadequate, such as measuring multiple biological pathways simultaneously, or revealing off-target drug effects. HCA has stepped into this void by demonstrating how particular proteins are affected by the application of a molecule to the cell line of interest. | |
| HCA is also used as a secondary screen to reveal potential toxicities. While detecting structural and biochemical changes in cells is vital to the discovery and development of new pharmaceuticals, it is also critical in measuring their toxicity during the early stages of drug screening. Automating the analysis of images of cells allows drug companies to use "high content screening" to measure subtle but important changes both rapidly and accurately. | |
| A new study conducted by a joint U.S. - Irish team has now made the first step toward the development of both a fast and fairly comprehensive method of screening of biological activity and cytotoxicity of nanoparticles. | |
| "We see two important factors that necessitate the development of such protocols" Dr. Nicholas Kotov tells Nanowerk. "Firstly, the synthesis of nanoparticles is much simpler than the synthesis of proteins and other drugs. Since minor changes in the synthetic protocol are likely to affect their interactions with cells, one can expect to see a tremendous surge of potential candidates for toxicity/biological activity screening. Secondly, considering the diversity of nanoparticles being synthesized, one needs a unified approach for screening nanomaterials. Such a systematic approach is not only fundamental to the construction of a unified database for biological and cytotoxic effects of nanomaterials but will also enable scientists to synthesize safer and more efficacious nanostructures at an ever-more efficient rate." | |
| Kotov, a Professor in Chemical Engineering at the University of Michigan, in collaboration with the research group of Dr. Yurii Gun'ko from the Trinity College Dublin, Ireland, says that we can expect the engineering of biologically functional nanostructures to follow the path of synthetic pharmaceuticals in drug discovery: "In the near future, panels of nanoparticles with slightly varying properties and structures will be synthesized and evaluated for cytotoxicity before qualified candidates are designated subsequent developments." | |
| In a paper in the May 10, 2008 online edition of ACS Nano, the researchers describe their universal approach to the investigation of toxicity of nanoparticles and other nanocolloids ("High-Content Screening as a Universal Tool for Fingerprinting of Cytotoxicity of Nanoparticles"). This method in effect suggests that the treatment of nanoparticles in respect to cells would be similar to that of drug candidates, for evaluation of which HCA is widely used. | |
| Kotov says that the team's motivation was to accelerate and systematize the process of assessing the effects of nanoparticles on cells. "There are so many parameters of both cells and nanoparticles that can effect toxicity. One has to screen the nanoparticles in respect to size, core material, composition of the stabilizer shell, length of exposure, and concentration. HCA is a marriage between a computer and confocal microscopy, and thus, it offers substantial advantages in the form of automated screening and uniform processing of the results. In the end, one can quickly obtain 'fingerprints' of the toxic effects of nanocolloids in respect to numerous characteristics." | |
| In future, the multiparametric nature of these fingerprints – or profiles – will allow cytotoxicity analyses to be conducted at much higher throughput and accuracy. Kotov points out that the application of HCA technology in the study of nanomaterials is not limited to colloidal nanoparticles and cytotoxicity studies, as the team already is in the process of demonstrating its novel use on biocompatibility assessment of multilayer thin films produced from a layer-by-layer assembly. | |
 |
|
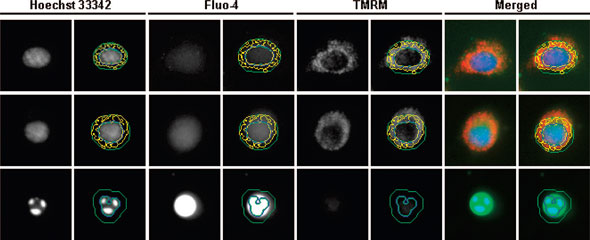
| Representative fluorescence images of a healthy (first row), an impaired (middle row), and a dying (last row) cell acquired and processed by the IN Cell HCS system. The nucleus, stained by Hoechst 3342, is outlined by a blue circle. The cell body or the intracellular region is enclosed by a green circle, within which the intensity of Fluo-4 fluorescence is measured. The punctuate, TMRM-bound mitochondrial organelles in the cytosol are identified by the yellow inclusions. In the merged images, Hoechst 3342, Fluo-4, and TMRM stains are shown in blue, green, and red, respectively. As the health condition of cells deteriorates, the nucleus shrinks and becomes fragmented, the Fluo-4 stain intensifies and signals a sharp increase in intracellular free calcium concentration, and the TMRM stain diminishes as a result of reduction in mitochondrial membrane potential. (Reprinted with permission from American Chemical Society) | |
| While understanding the detailed mechanism of the toxic effects of nanoparticles is certainly one obvious application for this technique, others include studies of anticancer formulations based on nanoparticles. A recent example of such an application is gold particles with antileukemia drug attached to the surfaces as described in one of Kotov's recent publications ("Gold Nanoparticles Enhance the Anti-Leukemia Action of a 6-Mercaptopurine Chemotherapeutic Agent"). | |
| In addition to adapting the HCA technique to nanoparticulate material, the researchers also report two other interesting findings: "Most importantly, we demonstrated that undifferentiated and differentiated cells respond differently to quantum dot (cadmium telluride) induced cytotoxicity" says Kotov. "Specifically, the differentiated cells are more sensitive and vulnerable to quantum dot treatment, which can be understood as the demonstration of adaptability of cells in undifferentiated state. This difference should be taken into account in the establishment of treatment dosage for any NP-based biological studies or therapies." | |
| The scientists are also exploring the use of HCA technology to study the transport and localization of engineered nanoparticles in living cells by proposing a broader change in the way nanoparticles' effect on cells are evaluated. | |
| "HCA is used for screening drugs and by using the method, we basically open a wide field of subtle interactions of nanoparticles with cells, which eventually can lead to new advances in medicine. We believe this endeavor ultimately will help to shed light on the development of new drugs and drug delivery strategies based on nanomaterials." | |
| Kotov is quick to stress the limitations of traditional ex vivo nanoparticle toxicity tests related to the two-dimensional nature of cell cultures: "Cellular response in 2D cell cultures was shown to be different than cells in the natural tissue environment. More adequate 3D cell culture techniques – giving much better representation of the actual processes taking place in human tissues – need to be developed. HCA does afford analysis of 3D images in a similar way as we presented here, however, the 3D approach will indeed require development of appropriate 3D matrixes (scaffolds), cell culture techniques, and refinement of software algorithms for image analysis." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
