| Posted: Jun 26, 2006 | |
Carbon nanotubes as nanoextruders |
|
| (Nanowerk Spotlight) The ultimate internal pressure (∼40 GPa) that carbon nanotubes (CNTs) can resist is only an order of magnitude below the pressure in the center of Earth. Using this high strength against internal pressure, researchers have used CNTs as pressure cells for the deformation of crystalline materials. Controlled irradiation of multiwalled CNTs can cause large pressure buildup within the nanotube cores that can plastically deform, extrude, and break solid materials that are encapsulated inside the core. Carbon nanotubes thus offer a template for use as compression/extrusion cells to study pressure-induced phase transformations and deformations of various solid nanomaterials. | |
| Leading to these findings was research by Professor Florian Banhart at the University of Mainz in Germany, that have shown that the high strength of nanotubes persists even when carbon atoms in the graphitic layers are missing. The experiments have been carried out in a high-resolution electron microscope where carbon atoms were knocked from the graphitic shells by an energetic electron beam. It was found that nanotubes shrink under electron irradiation but are not destroyed. | |
| This observation came as a surprise but has meanwhile been explained by computer simulations. Vacancies in the graphite lattice (open sites which are left after the displacement of carbon atoms) have an unexpectedly high mobility at the temperature of this experiment (600°C) and coalesce to form divacancies. Divacancies are unstable, however, and collapse by closing open bonds so that a coherent graphite layer is restored. Such a layer does not just consist of hexagonal rings only as the perfect graphite lattice, but also of pentagonal or heptagonal rings. | |
| Hence, the graphite structure is able to heal itself when atoms are missing, and its high mechanical stability is preserved. However, the surface area is getting smaller so that cylindrically closed graphite shells contract under electron irradiation. | |
| If other materials are encapsulated inside CNTs while irradiated with electrons, counterpressure from inside is exerted against the contraction. During the experiments in Mainz, it has been observed that the contraction is so strong that the encapsulated crystals, e.g., metals or metal carbides, are massively deformed and finally squeezed through the tubes in the axial direction. | |
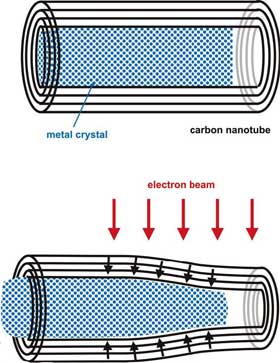 |
Principle of the experiment: Electron irradiation of an initially perfect CNTs filled with a crystalline metal nanowire leads to a collapse of the tube. The shrinking tube deforms the encapsulated metal crystal. (Source: University of Mainz) |
| Calculations show that pressures up to 400 000 atmospheres can be built up in the radial direction inside the collapsing tubes during such an experiment. This is by far enough to deform even hard materials. | |
| Based on these findings, an international research group - Banhart's group in Mainz, the Helsinki University of Technology, Finland, the Advanced Materials Group at IPICyT in San Luis Potosi, Mexico and the Rensselaer Polytechnic Institute in the U.S. - used CNTs as "nanolaboratories" to study the deformation of individual metal crystals. Their findings were reported in an article titled "Carbon Nanotubes as High-Pressure Cylinders and Nanoextruders", published in the May 26, 2006 issue of Science. | |
| One advantage of the present experiment is that individual nanometer-sized crystals can now be studied in situ and deformed under high pressure, although irradiation effects in the crystals might occur and have to be considered. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
