| Posted: Sep 25, 2006 | |
Stealth nanoparticles for long term in vivo drug treatment |
|
| (Nanowerk Spotlight) Phagocytosis is a cellular phenomena that describes the process in which phagocytes (specialized cells such as macrophages) destroy viruses and foreign particles in blood. Phagocytes are an important part of the immune system. Unfortunately, phagocytes are also a major limitation for the intravenous delivery of polymeric nanoparticles. The use of such nanoparticles to deliver therapeutic agents is currently being studied as a promising method by which drugs can be effectively targeted to specific cells in the body, such as cancerous cells. Researchers at Penn State are trying to trick the body's immune system, and increase the circulation time of nano drug carriers in the blood, with stealth drug nanoparticles that could be fabricated by self-assembling a shell on the surface of a solid drug core. This research could lead to the possibility of long term drug treatment in vivo. | |
| Nanoparticles become recognizable to the cells of the mononuclear phagocyte system (MPS), and are subsequently cleared from circulation by phagocytosis, through a process called opsonization. An opsonin is a proteinaceous molecule that acts as a binding enhancer for the process of phagocytosis. Phagocytic cells express receptors that bind opsonin molecules. | |
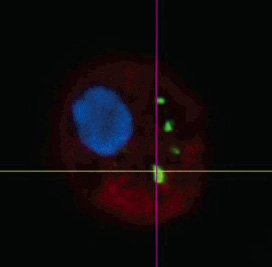 | |
| Uptake of nanoparticles in a macrophage cell. (Reprinted with permission from Institute of Physics Publishing) | |
| In order to improve the in vivo survival rate of nanoparticles, researchers are therefore trying to engineer a long circulating nanocarrier with a surface that can avoid opsonin adsorption and the subsequent clearance from the body by phagocytic cells. | |
| Dr. Michael V. Pishko, Professor of Chemical Engineering and Materials Science and Engineering at Penn State, explained the background of his group's research into nanoparticulate drug delivery to Nanowerk: "It has been established that the physicochemical characteristics of a polymeric nanoparticle such as surface charge, size, functional groups, and hydrophilicity can affect its uptake by the cells of the MPS. Previously, it was suggested that a long-circulating, "stealth" carrier could be designed by incorporating hydrophilic polymers on the surface of the polymeric carrier." | |
| It was found that a neutral and hydrophilic surface can lead to a longer half-life in the blood circulation. | |
| "In particular, nanoparticles whose surfaces were modified by the incorporation of poly(ethylene glycol) (PEG) during nanoparticle formulation have illustrated a decreased uptake by cells of the MPS and an increased circulation time to effectively target diseased cells" says Pishko. | |
| PEG molecules on the surface of a polymeric nanoparticle can reduce the adsorption of opsonins and other serum proteins by a mechanism known as the "steric repulsion effect". It has been hypothesized that PEG molecules on the surface of a nanoparticle will form a dynamic molecular "cloud" over the particle surface. This cloud of mobile and flexible PEG molecules imparts a repulsive effect by which it is energetically unfavorable for proteins to adsorb to PEG molecules. | |
| "For our study, we used a layer-by-layer (LbL) assembly process to produce core-shell polymeric nanoparticles where fluorescent nanobeads (to simulate a solid drug core) were encapsulated in macromolecular nanoshells" explains Pishko. "The nanoshell composed of multilayered polyelectrolytes was modified with PEG of varying molecular weights (either 2000, 5000, or 20 000 Da) to form a hydrophilic and long-circulating nanoparticle." | |
| The researchers determined a relationship between uptake and physicochemical properties including surface charge and the hydrophilicity of the core-shell nanoparticles. | |
| "We were able to determine that a low negative to neutral and hydrophilic nanoshell can reduce uptake of coreshell nanoparticles in vitro" Pishko concludes. | |
| The findings are published in a paper, titled "Macrophage Uptake of Core-Shell Nanoparticles Surface Modified with Poly(ethylene glycol)", which appeared in the August 1, 2006 online edition of Langmuir. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com.
