| Posted: Oct 05, 2006 | |
A new spin on an older technology |
|
| (Nanowerk Spotlight) Because of the huge effective surface area, the ability to blend different types of polymers, and the fact that the process is conducted at room temperature so that biological compounds can be loaded into the fibers, electrospinning has enormous potential to create new families of higher performance products across a wide array of industry sectors. For a technique invented in 1934, we are just now beginning to see its true potential. | |
| Electrospinning may be a well established method for creating small diameter fibers, but its true potential for performance improvements across a wide range of applications has only recently been recognized. | |
| This breakthrough nanotechnology has potential to create new industry models in manufacturing and supply for medicine, industrial filtration, drug delivery, and the military. Applications for electrospun fibers range from scaffolding for tissue engineering (bone, cartilage blood vessel regeneration), to bioresorbable drug delivery systems for hard-to-treat wounds, to protective clothing with reactive sites for biological recognition elements (chemical and bio-hazard gear). | |
| Basically, electrospinning uses an electrical charge to form a mat of extremely fine fibers which have diameters typically between 20 and 200 nm. A polymer, sol-gel or composite solution (or melt) is loaded into a syringe tipped with a metallic needle. A high-voltage power supply is applied between the needle and a grounded collector plate. As the voltage is applied, and the polymer is forced from the syringe through the tip, the initial droplet is stretched into a structure known as a Taylor Cone. If the polymer’s viscosity is sufficiently high, and if stream breakup does not occur, then an electrified liquid jet of polymer is formed and spirals downward toward the collector at roughly 300 mph. As the solvent evaporates, electrostatic repulsion accelerates and elongates the jet into long whipping fibers that form a matted pile on a grounded receiving screen 10 to 30 cm away. These charged polymeric fibers can be collected in sheets, tubes, and complex geometrical shapes. (See Figure 1) | |
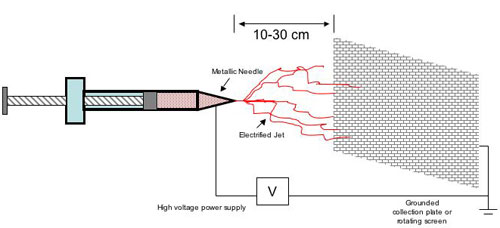 |
|
| Figure 1 (Source: Jim Clements) | |
| The electrospinning process was invented and patented by Antonin Formhals in 1934, with dozens of additional patents filed on melts and solvent systems over the last 72 years. Renewed attention to the technique increased in the mid-90’s largely due to overall interest in nanomaterials. | |
| Reports in relevant literature cite more than 100 synthetic and natural polymers which have been successfully electrospun, including PP, PE, PA, PVA, elastin, collagen, DNA and other biomaterials. However, the most recent advances in electrospinning have resulted from a focus to control the parameters of the process. Because the process itself is impacted by so many variables, and the interrelationship between the parameters is so complex, the diffusion of electrospinning as a commercial process has been restricted. | |
| One recent example of improved control is from Dr. Liwei Lin’s lab at UC, Berkeley. Along with visiting professor Daoheng Sun, Lin was able to refine the process to create an orderly and directed pattern of electrospun nanofibers, overcoming a key obstacle in the process. Termed ‘near-field electrospinning’ their innovation is basically a four-step improvement which includes the use of a fine-tipped tungstun electrode in place of a needle-nozzle, a reduction in the distance between tip and collector from 30cm to 0.5-3.0 mm, and a decrease in applied voltage from 30kV to 600V. | |
| Possible near-term applications for near-field electrospinning include biosensors which require precise placement of fibers, as well as tissue-scaffolding using non-woven fabrics with defined pattern characteristics. Work on refining this process continues. | |
| Other Advances: | |
| One key attribute of electrospinning is its ability to create three dimensional structures. In 2006, engineers and scientists led by Dr. Gary Bowlin from Virginia Commonwealth University, designed and electrospun a fiber to be used for vascular grafts. Using a blend of polydioxanone (PDO), a synthetic polymer typically used as suture material, and elastin, a natural polymer present in arterial walls, the researchers were able to manipulate the fiber into a hose-like structure for use as a graft. | |
| One potential benefit is that a graft made of this polymer blend would promote tissue regeneration and in-growth, while significantly limiting the inflammatory response and poor wound healing that sometimes occurs with conventional graft materials such as PTFE. | |
| By James T. Clements, Copyright Nanowerk LLC | |
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
