 Scientists have used a suite of correlative, multimodal microscopy methods to visualise, for the first time, why perovskite materials are seemingly so tolerant of defects in their structure.
Scientists have used a suite of correlative, multimodal microscopy methods to visualise, for the first time, why perovskite materials are seemingly so tolerant of defects in their structure.
Nov 22nd, 2021
Read more
 New approach solves a persistent problem of intermittency that has hindered use of the tiny light emitters for biological imaging or quantum photonics.
New approach solves a persistent problem of intermittency that has hindered use of the tiny light emitters for biological imaging or quantum photonics.
Nov 22nd, 2021
Read more
 Researchers have developed a combined electric current 2D material sensor for the detection of breast cancer cells. This ultra-sensitive sensor was able to detect electrical signals from a record low number of cancer cells.
Researchers have developed a combined electric current 2D material sensor for the detection of breast cancer cells. This ultra-sensitive sensor was able to detect electrical signals from a record low number of cancer cells.
Nov 22nd, 2021
Read more
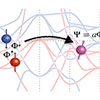 For the first time through experiments and theory, researchers have been able to measure spin mixing in a ferromagnetic material. Through the experimental measurements, they discovered that a common factor in spin equations, in common use since the 1950s, has been significantly underestimated.
For the first time through experiments and theory, researchers have been able to measure spin mixing in a ferromagnetic material. Through the experimental measurements, they discovered that a common factor in spin equations, in common use since the 1950s, has been significantly underestimated.
Nov 22nd, 2021
Read more
 Long-range magnetic order has been observed in quasicrystals, strange solids that show forbidden crystal symmetries, for the first time.
Long-range magnetic order has been observed in quasicrystals, strange solids that show forbidden crystal symmetries, for the first time.
Nov 22nd, 2021
Read more
 Researchers have developed a human nuclear transfer stem cell-based microrobot for minimally invasive delivery into the brain tissue via the intranasal pathway.
Researchers have developed a human nuclear transfer stem cell-based microrobot for minimally invasive delivery into the brain tissue via the intranasal pathway.
Nov 22nd, 2021
Read more
 With the power of nanotechnology, investigators have discovered that cancer cells strengthen by forming nanotubes that they use to suck mitochondria out of immune cells.
With the power of nanotechnology, investigators have discovered that cancer cells strengthen by forming nanotubes that they use to suck mitochondria out of immune cells.
Nov 22nd, 2021
Read more
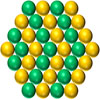 Nickel-cobalt metal dimer on nitrogen-doped carbon can catalyze electrolysis under both acidic and basic conditions.
Nickel-cobalt metal dimer on nitrogen-doped carbon can catalyze electrolysis under both acidic and basic conditions.
Nov 19th, 2021
Read more
 Demonstrating that a material thought to be always chemically inert, hexagonal boron nitride, can be turned chemically active holds potential for a new class of catalysts with a wide range of applications, according to an international team of researchers.
Demonstrating that a material thought to be always chemically inert, hexagonal boron nitride, can be turned chemically active holds potential for a new class of catalysts with a wide range of applications, according to an international team of researchers.
Nov 19th, 2021
Read more
 Special ferroelectric features offer promise for microelectronics and energy applications.
Special ferroelectric features offer promise for microelectronics and energy applications.
Nov 19th, 2021
Read more
 Researchers are investigating the use of whole-cell vaccines to fight urinary tract infection (UTI), part of an effort to tackle the increasingly serious issue of antibiotic-resistant bacteria.
Researchers are investigating the use of whole-cell vaccines to fight urinary tract infection (UTI), part of an effort to tackle the increasingly serious issue of antibiotic-resistant bacteria.
Nov 19th, 2021
Read more
 Scientists obtain new structural insights into an important class of thermoelectric materials that until now has evaded detailed characterization.
Scientists obtain new structural insights into an important class of thermoelectric materials that until now has evaded detailed characterization.
Nov 19th, 2021
Read more
 The ability to take spectra from individual molecules promises to be a vital addition to the toolkit of researchers looking at excited molecules.
The ability to take spectra from individual molecules promises to be a vital addition to the toolkit of researchers looking at excited molecules.
Nov 19th, 2021
Read more
 A method for predicting exotic states of matter could be useful for processing quantum information.
A method for predicting exotic states of matter could be useful for processing quantum information.
Nov 19th, 2021
Read more
 Nanoplastics is a new type of environmental pollutant. Its particles can easily enter the bodies of animals through diet and breathing, and affect the physiological functions of organisms.
Nanoplastics is a new type of environmental pollutant. Its particles can easily enter the bodies of animals through diet and breathing, and affect the physiological functions of organisms.
Nov 19th, 2021
Read more
 Researchers developed battery-like electrochemical Nb2CTx MXene electrodes with stable voltage output and high energy density by using a high-voltage scanning strategy. These latest findings may lead to a breakthrough in inventing the powerful battery of the next generation.
Researchers developed battery-like electrochemical Nb2CTx MXene electrodes with stable voltage output and high energy density by using a high-voltage scanning strategy. These latest findings may lead to a breakthrough in inventing the powerful battery of the next generation.
Nov 18th, 2021
Read more
 Scientists have used a suite of correlative, multimodal microscopy methods to visualise, for the first time, why perovskite materials are seemingly so tolerant of defects in their structure.
Scientists have used a suite of correlative, multimodal microscopy methods to visualise, for the first time, why perovskite materials are seemingly so tolerant of defects in their structure.















 Subscribe to our Nanotechnology News feed
Subscribe to our Nanotechnology News feed