Researchers synthesize bilayer borophene
 Scientists report the synthesis of large-size, single-crystalline bilayer borophene on the Cu(111) surface by molecular beam epitaxy.
Scientists report the synthesis of large-size, single-crystalline bilayer borophene on the Cu(111) surface by molecular beam epitaxy.
Nov 15th, 2021
Read more
 Scientists report the synthesis of large-size, single-crystalline bilayer borophene on the Cu(111) surface by molecular beam epitaxy.
Scientists report the synthesis of large-size, single-crystalline bilayer borophene on the Cu(111) surface by molecular beam epitaxy.
Nov 15th, 2021
Read more This novel design uses a straight grating antenna, which allowed a small amount of light to scatter out of the plane at the designed angle, while the rest could also pass through. The grating was made of silicon waveguides coated with silicon dioxide.
This novel design uses a straight grating antenna, which allowed a small amount of light to scatter out of the plane at the designed angle, while the rest could also pass through. The grating was made of silicon waveguides coated with silicon dioxide.
Nov 15th, 2021
Read more The project NanoQI will be the first consortium to optimize and multimodally combine X-ray diffraction analysis (XRD) and X-ray reflectometry (XRR) with novel hyperspectral imaging (HSI) technology to provide industry access to real-time assessment of nano-dimensions, structure and morphology of thin film nanomaterials, as well as correlative imaging of their homogeneity and defects.
The project NanoQI will be the first consortium to optimize and multimodally combine X-ray diffraction analysis (XRD) and X-ray reflectometry (XRR) with novel hyperspectral imaging (HSI) technology to provide industry access to real-time assessment of nano-dimensions, structure and morphology of thin film nanomaterials, as well as correlative imaging of their homogeneity and defects.
Nov 15th, 2021
Read more Researchers develop a new method for the generation of controllable electrical explosions.
Researchers develop a new method for the generation of controllable electrical explosions.
Nov 15th, 2021
Read more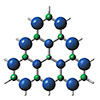 Researchers synthesize and crystallize a molecule that is otherwise too unstable to fully study in the laboratory, and is a model of a revolutionary class of magnets.
Researchers synthesize and crystallize a molecule that is otherwise too unstable to fully study in the laboratory, and is a model of a revolutionary class of magnets.
Nov 15th, 2021
Read more Researchers have found a cheaper, easier way to create a nanoscopic forest of carbon nanotubes, a tiny material that could pack a big punch when it comes to protecting medical implants in the human body.
Researchers have found a cheaper, easier way to create a nanoscopic forest of carbon nanotubes, a tiny material that could pack a big punch when it comes to protecting medical implants in the human body.
Nov 15th, 2021
Read more Scientists suggest a method to synthesize borophene, the 2D version of boron, in a way that could make it easier to free up or manipulate.
Scientists suggest a method to synthesize borophene, the 2D version of boron, in a way that could make it easier to free up or manipulate.
Nov 12th, 2021
Read more Researchers have shown that liquid metals can offer their characteristics to the pharmaceutical and chemical industries, with the possible elimination of moving parts in continuous flow reactors providing improved performance and reduced maintenance costs.
Researchers have shown that liquid metals can offer their characteristics to the pharmaceutical and chemical industries, with the possible elimination of moving parts in continuous flow reactors providing improved performance and reduced maintenance costs.
Nov 12th, 2021
Read more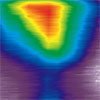 Researchers have combined two traditional semiconductor doping methods to achieve new efficiencies in the topological insulator bismuth-selenide.
Researchers have combined two traditional semiconductor doping methods to achieve new efficiencies in the topological insulator bismuth-selenide.
Nov 12th, 2021
Read more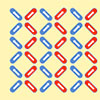 Researchers studied microscopic arrays of nanomagnets that provide the first experimental demonstration of a classic theoretical model, known as the 'one-dimensional random field Ising model'.
Researchers studied microscopic arrays of nanomagnets that provide the first experimental demonstration of a classic theoretical model, known as the 'one-dimensional random field Ising model'.
Nov 12th, 2021
Read more Chemical engineers have overcome a problem in the manufacturing of perovskites that reduces their efficiency as solar panels. The approach produced perovskite solar panels with an efficiency of 23.9% and operational stability longer than 1000?hours.
Chemical engineers have overcome a problem in the manufacturing of perovskites that reduces their efficiency as solar panels. The approach produced perovskite solar panels with an efficiency of 23.9% and operational stability longer than 1000?hours.
Nov 12th, 2021
Read more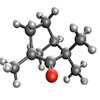 Scientists report on a newly-developed machine learning algorithm for LIED to extract the three-dimensional structure of large and complex molecules.
Scientists report on a newly-developed machine learning algorithm for LIED to extract the three-dimensional structure of large and complex molecules.
Nov 12th, 2021
Read more Researchers have developed a silicon coating that, when applied to the surface of a glass lens, can counteract the effects of dispersion.
Researchers have developed a silicon coating that, when applied to the surface of a glass lens, can counteract the effects of dispersion.
Nov 11th, 2021
Read more The project will involve 14 Early Stage Researchers, who have been selected to develop a novel technological platform for photonics based on an emerging class of semiconductors, metal-halide perovskites, with exceptional optical and electrical properties.
The project will involve 14 Early Stage Researchers, who have been selected to develop a novel technological platform for photonics based on an emerging class of semiconductors, metal-halide perovskites, with exceptional optical and electrical properties.
Nov 11th, 2021
Read more Molecular self-assembly expert Chun-Long Chen describes the challenges and opportunities ahead in bio-inspired materials design.
Molecular self-assembly expert Chun-Long Chen describes the challenges and opportunities ahead in bio-inspired materials design.
Nov 11th, 2021
Read more Researchers demonstrate a technique that tracks Brownian motion in three dimensions and characterization of individual nanoparticles using a fiber-based high-finesse microcavity.
Researchers demonstrate a technique that tracks Brownian motion in three dimensions and characterization of individual nanoparticles using a fiber-based high-finesse microcavity.
Nov 11th, 2021
Read more