Engineers produce a fisheye lens that's completely flat
 The single piece of glass produces crisp panoramic images.
The single piece of glass produces crisp panoramic images.
Sep 18th, 2020
Read more
 The single piece of glass produces crisp panoramic images.
The single piece of glass produces crisp panoramic images.
Sep 18th, 2020
Read more Researchers invent a way to explore the structures of functional optical materials.
Researchers invent a way to explore the structures of functional optical materials.
Sep 18th, 2020
Read more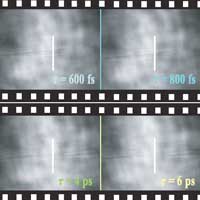 New all-optical imaging technique with an unprecedented frame rate allows scientists to visualize ultrafast transient phenomena.
New all-optical imaging technique with an unprecedented frame rate allows scientists to visualize ultrafast transient phenomena.
Sep 17th, 2020
Read more Automatic approach could enable precision fabrication of optical components and multimaterial structures.
Automatic approach could enable precision fabrication of optical components and multimaterial structures.
Sep 17th, 2020
Read more Researchers demonstrate a highly flexible but sturdy wearable piezoelectric harvester using the simple and easy fabrication process of hot pressing and tape casting.
Researchers demonstrate a highly flexible but sturdy wearable piezoelectric harvester using the simple and easy fabrication process of hot pressing and tape casting.
Sep 17th, 2020
Read more Researchers have tested whether gold nanoparticles could be toxic to cells, finding how they affect lipid membranes depends on their size.
Researchers have tested whether gold nanoparticles could be toxic to cells, finding how they affect lipid membranes depends on their size.
Sep 17th, 2020
Read more Researchers have created a smart drug delivery system that reduces inflammation in damaged nervous tissues and may help treat spinal cord injuries and other neurological disorders.
Researchers have created a smart drug delivery system that reduces inflammation in damaged nervous tissues and may help treat spinal cord injuries and other neurological disorders.
Sep 17th, 2020
Read more Scientists show that high-resolution movies of molecular dynamics can be recorded using electrons ejected from the molecule by an intense laser field.
Scientists show that high-resolution movies of molecular dynamics can be recorded using electrons ejected from the molecule by an intense laser field.
Sep 17th, 2020
Read more Researchers have discovered that colloidal rods can be moved on a chip quickly, precisely, and in different directions, almost like chess pieces. A pre-programmed magnetic field even enables these controlled movements to occur simultaneously.
Researchers have discovered that colloidal rods can be moved on a chip quickly, precisely, and in different directions, almost like chess pieces. A pre-programmed magnetic field even enables these controlled movements to occur simultaneously.
Sep 17th, 2020
Read more With a size 100 times smaller than an average human hair, the new detector can visualize features that are much smaller than previously possible, leading to what is known as super-resolution imaging.
With a size 100 times smaller than an average human hair, the new detector can visualize features that are much smaller than previously possible, leading to what is known as super-resolution imaging.
Sep 17th, 2020
Read more Adding calcium to graphene creates an extremely promising superconductor, but where does the calcium go? In a new study, researchers have for the first time confirmed what actually happens to those calcium atoms.
Adding calcium to graphene creates an extremely promising superconductor, but where does the calcium go? In a new study, researchers have for the first time confirmed what actually happens to those calcium atoms.
Sep 17th, 2020
Read more Researchers have artificially reproduced a nanoscale coating on different types of surfaces that usually covers the eyes of fruit flies, and which provides anti-reflective, anti-adhesive properties.
Researchers have artificially reproduced a nanoscale coating on different types of surfaces that usually covers the eyes of fruit flies, and which provides anti-reflective, anti-adhesive properties.
Sep 16th, 2020
Read more Researchers have found a completely new method to check the electronic properties of oxide materials. This opens the door to even tinier components and perhaps more sustainable electronics.
Researchers have found a completely new method to check the electronic properties of oxide materials. This opens the door to even tinier components and perhaps more sustainable electronics.
Sep 16th, 2020
Read more Researchers have cleared the obstacle that had prevented the creation of electrically driven nanolasers for integrated circuits. The approach enables coherent light source design on the scale not only hundreds of times smaller than the thickness of a human hair but even smaller than the wavelength of light emitted by the laser.
Researchers have cleared the obstacle that had prevented the creation of electrically driven nanolasers for integrated circuits. The approach enables coherent light source design on the scale not only hundreds of times smaller than the thickness of a human hair but even smaller than the wavelength of light emitted by the laser.
Sep 16th, 2020
Read more Scientists have uncovered a link between the microscopic movements of particles in a liquid and its ability to absorb heat.
Scientists have uncovered a link between the microscopic movements of particles in a liquid and its ability to absorb heat.
Sep 16th, 2020
Read more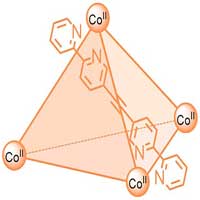 Research team develops new NMR methods for investigating paramagnetic complexes and supramolecular cages.
Research team develops new NMR methods for investigating paramagnetic complexes and supramolecular cages.
Sep 16th, 2020
Read more