 Physicists have demonstrated the real possibility of using superconductor/ferromagnet systems to create magnonic crystals, which will be at the core of spin-wave devices to come in the post-silicon era of electronics.
Physicists have demonstrated the real possibility of using superconductor/ferromagnet systems to create magnonic crystals, which will be at the core of spin-wave devices to come in the post-silicon era of electronics.
Aug 21st, 2019
Read more
 Researchers have developed artificial 'chameleon skin' that changes colour when exposed to light and could be used in applications such as active camouflage and large-scale dynamic displays.
Researchers have developed artificial 'chameleon skin' that changes colour when exposed to light and could be used in applications such as active camouflage and large-scale dynamic displays.
Aug 21st, 2019
Read more
 A team of researchers has developed a molecular propeller that enables unidirectional rotations on a material surface when energized.
A team of researchers has developed a molecular propeller that enables unidirectional rotations on a material surface when energized.
Aug 20th, 2019
Read more
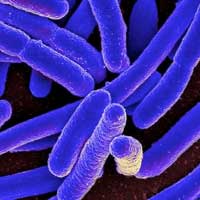 A team of researchers has discovered a way to find and beat superbugs, providing a critical breakthrough against many deadly infectious diseases.
A team of researchers has discovered a way to find and beat superbugs, providing a critical breakthrough against many deadly infectious diseases.
Aug 20th, 2019
Read more
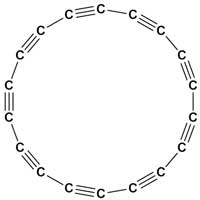 Scientists have stabilized and imaged a ring of 18 carbon atoms for the first time.
Scientists have stabilized and imaged a ring of 18 carbon atoms for the first time.
Aug 20th, 2019
Read more
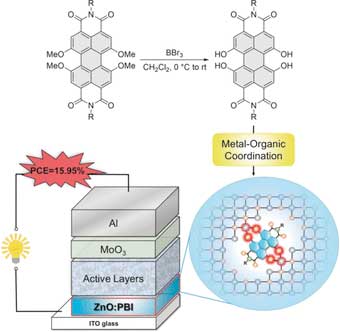 Organic dye in zinc oxide interlayer stabilizes and boosts the performance of organic solar cells.
Organic dye in zinc oxide interlayer stabilizes and boosts the performance of organic solar cells.
Aug 20th, 2019
Read more
 Researchers have developed and tested a novel approach for tackling melanoma.
Researchers have developed and tested a novel approach for tackling melanoma.
Aug 20th, 2019
Read more
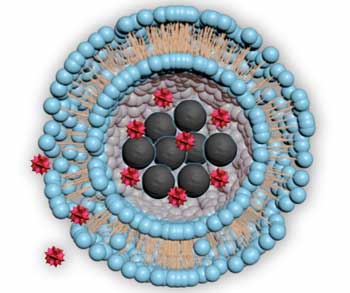 Magnetic particles allow drugs to be released at precise times and in specific areas.
Magnetic particles allow drugs to be released at precise times and in specific areas.
Aug 20th, 2019
Read more
 Researchers want to recreate the chemistries found in natural materials and build similar structures in synthetic materials. By doing so, they can develop new, improved materials for use in sensors, healthcare applications, and much more.
Researchers want to recreate the chemistries found in natural materials and build similar structures in synthetic materials. By doing so, they can develop new, improved materials for use in sensors, healthcare applications, and much more.
Aug 20th, 2019
Read more
 Work could lead to the creation of smart watches with holographic displays, printed security cryptograms.
Work could lead to the creation of smart watches with holographic displays, printed security cryptograms.
Aug 20th, 2019
Read more
 Researchers have developed a skin patch with tiny needles that painlessly collect interstitial fluid (ISF) for testing.
Researchers have developed a skin patch with tiny needles that painlessly collect interstitial fluid (ISF) for testing.
Aug 19th, 2019
Read more
 Researchers show that a previously unexploited green macroalgae species could be used to extract cellulose nanofibers, which can then be formed into paper sheets with tailored pore size that are utilized for point-of-use water treatment.
Researchers show that a previously unexploited green macroalgae species could be used to extract cellulose nanofibers, which can then be formed into paper sheets with tailored pore size that are utilized for point-of-use water treatment.
Aug 19th, 2019
Read more
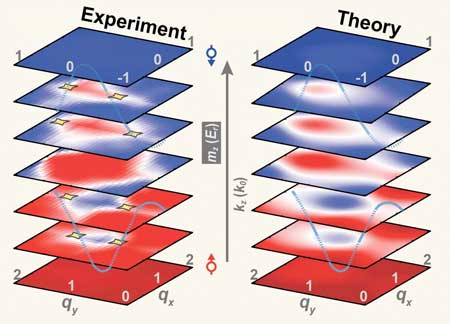 Physicists have successfully created the world's first 3D simulation of topological matter consisting of ultracold atoms.
Physicists have successfully created the world's first 3D simulation of topological matter consisting of ultracold atoms.
Aug 19th, 2019
Read more
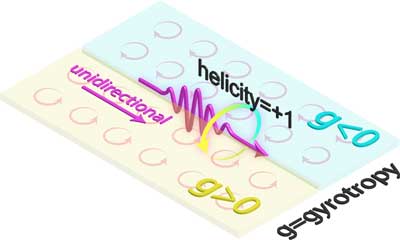 Researchers have created a quantum spin wave for light. This can be a carrier of information for future nanotechnologies but with a unique twist: they only flow in one direction.
Researchers have created a quantum spin wave for light. This can be a carrier of information for future nanotechnologies but with a unique twist: they only flow in one direction.
Aug 19th, 2019
Read more
 Chemists have created a new compound for flexible drug delivery that specifically targets prostate cancer cells. Incorporating four different molecules, the compound prevents tumour cells from multiplying, can be detected by medical imaging and has staying power in the bloodstream.
Chemists have created a new compound for flexible drug delivery that specifically targets prostate cancer cells. Incorporating four different molecules, the compound prevents tumour cells from multiplying, can be detected by medical imaging and has staying power in the bloodstream.
Aug 19th, 2019
Read more
 Engineers have developed experimental stickers that pick up physiological signals emanating from the skin, then wirelessly beam these health readings to a receiver clipped onto clothing. It's all part of a system called BodyNet.
Engineers have developed experimental stickers that pick up physiological signals emanating from the skin, then wirelessly beam these health readings to a receiver clipped onto clothing. It's all part of a system called BodyNet.
Aug 17th, 2019
Read more
 Physicists have demonstrated the real possibility of using superconductor/ferromagnet systems to create magnonic crystals, which will be at the core of spin-wave devices to come in the post-silicon era of electronics.
Physicists have demonstrated the real possibility of using superconductor/ferromagnet systems to create magnonic crystals, which will be at the core of spin-wave devices to come in the post-silicon era of electronics.















 Subscribe to our Nanotechnology News feed
Subscribe to our Nanotechnology News feed