'GO dough' makes graphene easy to shape and mold
 New form of graphene oxide is fun to play with -- and solves manufacturing challenges.
New form of graphene oxide is fun to play with -- and solves manufacturing challenges.
Jan 25th, 2019
Read more
 New form of graphene oxide is fun to play with -- and solves manufacturing challenges.
New form of graphene oxide is fun to play with -- and solves manufacturing challenges.
Jan 25th, 2019
Read more Temperature control of pore sizes in nanomaterials could lead to improved gas separation and storage applications.
Temperature control of pore sizes in nanomaterials could lead to improved gas separation and storage applications.
Jan 25th, 2019
Read more Scientists report a systematic study on the effects that using different forms of titanium oxide in planar perovskite solar cells has on the performance of the devices.
Scientists report a systematic study on the effects that using different forms of titanium oxide in planar perovskite solar cells has on the performance of the devices.
Jan 25th, 2019
Read more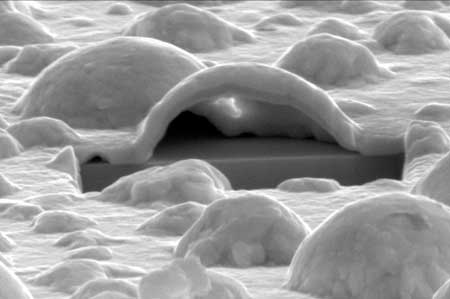 Technologically important noble metal oxidises more readily than expected.
Technologically important noble metal oxidises more readily than expected.
Jan 25th, 2019
Read more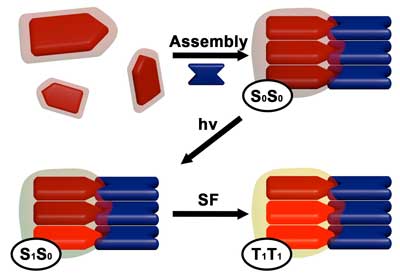 New nanomaterials may present a pathway to extra environment friendly and probably reasonably priced harvesting of photo voltaic vitality.
New nanomaterials may present a pathway to extra environment friendly and probably reasonably priced harvesting of photo voltaic vitality.
Jan 25th, 2019
Read more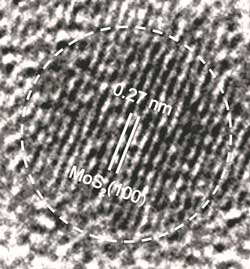 MoS2 quantum dots synthesised with the new approach can potentially generate cancer-killing properties.
MoS2 quantum dots synthesised with the new approach can potentially generate cancer-killing properties.
Jan 25th, 2019
Read more Nanocellulose offers renewable, biodegradable, strong and lightweight components for electronic products.
Nanocellulose offers renewable, biodegradable, strong and lightweight components for electronic products.
Jan 24th, 2019
Read more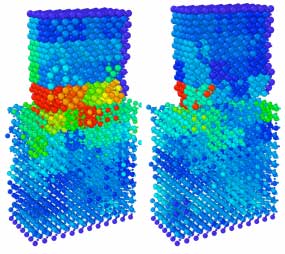 New research suggests that the cause for static electricity is tiny structural changes that occur at the surface of materials when they come into contact with each other. The finding could ultimately help technology companies create more sustainable and longer-lasting power sources for small electronic devices.
New research suggests that the cause for static electricity is tiny structural changes that occur at the surface of materials when they come into contact with each other. The finding could ultimately help technology companies create more sustainable and longer-lasting power sources for small electronic devices.
Jan 24th, 2019
Read more Researchers have developed a new method to finely tune adjacent layers of graphene to induce superconductivity. Their research provides new insights into the physics underlying this two-dimensional material's intriguing characteristics.
Researchers have developed a new method to finely tune adjacent layers of graphene to induce superconductivity. Their research provides new insights into the physics underlying this two-dimensional material's intriguing characteristics.
Jan 24th, 2019
Read more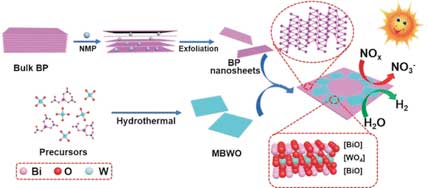 Active photocatalyst: 2D/2D heterojunction of black phosphorus and bismuth tungstate.
Active photocatalyst: 2D/2D heterojunction of black phosphorus and bismuth tungstate.
Jan 24th, 2019
Read more A team of engineering experts have pioneered a new way to ease production of van der Waals heterostructures with high-K dielectrics - assemblies of atomically thin two-dimensional crystalline materials.
A team of engineering experts have pioneered a new way to ease production of van der Waals heterostructures with high-K dielectrics - assemblies of atomically thin two-dimensional crystalline materials.
Jan 24th, 2019
Read more In the quest for smaller, faster 2D processors, researchers invent thermal lithography process for higher quality, lower cost, and mass production potential.
In the quest for smaller, faster 2D processors, researchers invent thermal lithography process for higher quality, lower cost, and mass production potential.
Jan 24th, 2019
Read more A newly developed graphene-based implant can record electrical activity in the brain at extremely low frequencies and over large areas, unlocking the wealth of information found below 0.1 Hz.
A newly developed graphene-based implant can record electrical activity in the brain at extremely low frequencies and over large areas, unlocking the wealth of information found below 0.1 Hz.
Jan 24th, 2019
Read more Tech wizards to tech novices may benefit from the ability to 'grow' solar cells and advance electronics, computers and energy conversion devices.
Tech wizards to tech novices may benefit from the ability to 'grow' solar cells and advance electronics, computers and energy conversion devices.
Jan 24th, 2019
Read more Scientists have discovered the relaxation dynamics of a zero-field state in skyrmions, a spinning magnetic phenomenon that has potential applications in data storage and spintronic devices.
Scientists have discovered the relaxation dynamics of a zero-field state in skyrmions, a spinning magnetic phenomenon that has potential applications in data storage and spintronic devices.
Jan 23rd, 2019
Read more Water splitting, the process of harvesting solar energy to generate energy-dense fuels, could be simplified thanks to new research.
Water splitting, the process of harvesting solar energy to generate energy-dense fuels, could be simplified thanks to new research.
Jan 23rd, 2019
Read more