A team of scientists from PML's Quantum Measurement Division has designed and tested a novel device that may lead to substantial progress in the new and fast-moving field of optomechanics.
Apr 10th, 2012
Read more
Lithium-sulfur batteries may be the power storage devices of the future. Newly developed porous nanoparticles containing sulfur deliver optimized battery performance.
Apr 10th, 2012
Read more
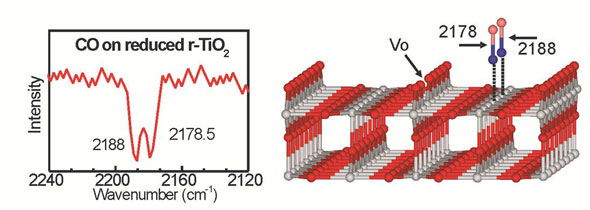 Scientists have developed a new infrared spectroscopy method in order to study processes at surfaces of oxides used as catalysts.
Scientists have developed a new infrared spectroscopy method in order to study processes at surfaces of oxides used as catalysts.
Apr 10th, 2012
Read more
The German Ministry of the Environment, Nature Protection and Nuclear Safety (BMU) has published a new report "Assessment tools for nanomaterials" ("Instrumente zur Bewertung von Nanomaterialien") which summarizes the discussion and results of the German NanoCommission's work and the Stakeholder Dialogue "Risk management in the nano world".
Apr 10th, 2012
Read more
The annual printed electronics award winners were announced at the IDTechEx Awards Dinner in Berlin, Germany on April 3rd. The awards recognize outstanding progress in the development and commercialization of printed electronics, an industry that produces a huge amount of technical innovation which will be used in many products.
Apr 10th, 2012
Read more
The Korea Advanced Institute of Science and Technology (KAIST) announced that a research team from the Department of Materials Science and Engineering has developed a technology that enables scientists and engineers to observe processes occurring in liquid media on the smallest possible scale which is less than a nanometer.
Apr 10th, 2012
Read more
 Researchers at the University of Central Florida have developed a novel technique that may give doctors a faster and more sensitive tool to detect pathogens associated with inflammatory bowel disease, including Crohn's disease.
Researchers at the University of Central Florida have developed a novel technique that may give doctors a faster and more sensitive tool to detect pathogens associated with inflammatory bowel disease, including Crohn's disease.
Apr 10th, 2012
Read more
Researchers successfully measured the exact status of the rapidly changing Helium atom using an atto second pulse. Thanks to this discovery, many ultrafast phenomena in nature can now be precisely measured.
Apr 9th, 2012
Read more
The problem with commercializing graphene that is synthesized onto metals over a wide area is that it can not be separated from the metal. However, a groundbreaking separation technology which is both cheap and environment friendly has been developed.
Apr 9th, 2012
Read more
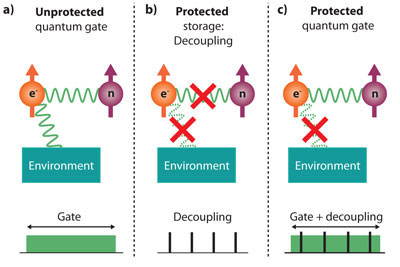 A team led by an Ames Laboratory scientist develops new technique for solid-state quantum information processing.
A team led by an Ames Laboratory scientist develops new technique for solid-state quantum information processing.
Apr 9th, 2012
Read more
A new study from Massachusetts General Hospital researchers finds that normalizing blood vessels within tumors, which improves the delivery of standard chemotherapy drugs, can block the delivery of larger nanotherapy molecules.
Apr 9th, 2012
Read more
Rice teams with MD Anderson, Baylor College of Medicine to explore drug and gene delivery.
Apr 9th, 2012
Read more
A North Carolina State University researcher has developed a more efficient, less expensive way of cooling electronic devices - particularly devices that generate a lot of heat, such as lasers and power devices.
Apr 9th, 2012
Read more
In receiving 2012 Barry M. Goldwater Scholarship, CNSE sophomore Sheila Smith is believed to be the first ever to be recognized in the field of nanotechnology.
Apr 9th, 2012
Read more
Scientists have discovered that when electric current is run through carbon nanotubes, objects nearby heat up while the nanotubes themselves stay cool, like a toaster that burns bread without getting hot.
Apr 9th, 2012
Read more
 A team led by University of Nebraska-Lincoln physicist Alexei Gruverman in collaboration with researchers in Spain and at the University of Wisconsin has discovered a significantly more efficient method of data storage that offers great promise for the future of technology.
A team led by University of Nebraska-Lincoln physicist Alexei Gruverman in collaboration with researchers in Spain and at the University of Wisconsin has discovered a significantly more efficient method of data storage that offers great promise for the future of technology.
Apr 9th, 2012
Read more





 Subscribe to our Nanotechnology News feed
Subscribe to our Nanotechnology News feed