Am 1. Dezember 2011 fand in Stuttgart ein Kongress zu Verbraucheraspekten im Umgang mit Nanotechnologien stat: Nano-Dialog Baden-Wuerttemberg - kleine Teilchen, grosse Fragen.
Dec 14th, 2011
Read more
Some molecules have unique and technologically useful optical properties; the medicinal properties of drugs depend on the direction of the twist; and within us twisted DNA can interact with different proteins. This twisting is called chirality and researchers at Case Western Reserve University have found they can use a macroscopic brute force to impose and induce a twist in an otherwise non-chiral molecule.
Dec 14th, 2011
Read more
Scientists are reporting development of a new cotton fabric that does clean itself of stains and bacteria when exposed to ordinary sunlight.
Dec 14th, 2011
Read more
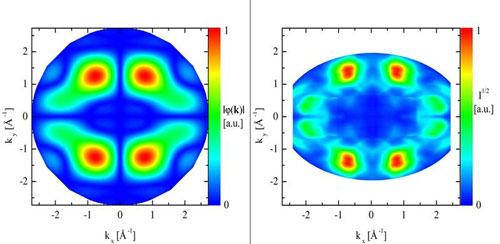 Einem Team von Physikern ist der Nachweis gelungen, dass auch die Elektronen in grossen Molekuelen - beispielsweise in organischen Halbleitern - mit hoher Praezision durch einzelne Orbitale beschrieben werden koennen.
Einem Team von Physikern ist der Nachweis gelungen, dass auch die Elektronen in grossen Molekuelen - beispielsweise in organischen Halbleitern - mit hoher Praezision durch einzelne Orbitale beschrieben werden koennen.
Dec 14th, 2011
Read more
 New detectors developed at MIT could provide easy visual identification of toxins or pathogens.
New detectors developed at MIT could provide easy visual identification of toxins or pathogens.
Dec 14th, 2011
Read more
 A new chemical technique for depositing a non-crystalline form of silicon into the long, ultra-thin pores of optical fibers has been developed by an international team of scientists.
A new chemical technique for depositing a non-crystalline form of silicon into the long, ultra-thin pores of optical fibers has been developed by an international team of scientists.
Dec 13th, 2011
Read more
Researchers at the Berkeley Lab have developed a promising new inexpensive technique for fabricating large-scale flexible and stretchable backplanes using semiconductor-enriched carbon nanotube solutions that yield networks of thin film transistors with superb electrical properties, including a charge carrier mobility that is dramatically higher than that of organic counterparts.
Dec 13th, 2011
Read more
A team of scientists have made it easier to study atomic or subatomic-scale properties of the building blocks of matter (which also include protons, neutrons and electrons) known as fermions by slowing down the movement of a large quantity of gaseous atoms at ultra-low temperature.
Dec 13th, 2011
Read more
At end of November, IDTechEx held the world's largest printed electronics and photovoltaics conference and tradeshow in Silicon Valley at the Santa Clara Convention Center.
Dec 13th, 2011
Read more
 A North Carolina State University invention has significant potential to improve the efficiency of solar cells and other technologies that derive energy from light.
A North Carolina State University invention has significant potential to improve the efficiency of solar cells and other technologies that derive energy from light.
Dec 13th, 2011
Read more
Analytical Pixels launched to design, manufacture, and commercialize analysis systems based on new concept of miniaturized chromatography.
Dec 13th, 2011
Read more
Research at Wayne State University, Mayo Clinic and Johns Hopkins offers potential treatment for macular degeneration and retinitis pigmentosa.
Dec 13th, 2011
Read more
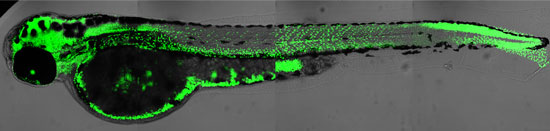 Researchers of the University of Zurich have discovered a new substance for labelling and visualization of DNA synthesis in whole animals. Applications for this technique include identifying the sites of virus infections and cancer growth, due to the abundance of DNA replication in these tissues. This approach should therefore lead to new strategies in drug development.
Researchers of the University of Zurich have discovered a new substance for labelling and visualization of DNA synthesis in whole animals. Applications for this technique include identifying the sites of virus infections and cancer growth, due to the abundance of DNA replication in these tissues. This approach should therefore lead to new strategies in drug development.
Dec 13th, 2011
Read more
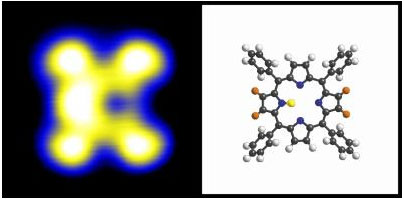 Physicists at the Technische Universitaet Muenchen have created a nano switch based on a single porphyrin ring. If one of two protons from the inside of the ring is removed, the remaining proton can take on any one of four positions, initiated by a single tunnel electron from the tip of a scanning tunneling microscope.
Physicists at the Technische Universitaet Muenchen have created a nano switch based on a single porphyrin ring. If one of two protons from the inside of the ring is removed, the remaining proton can take on any one of four positions, initiated by a single tunnel electron from the tip of a scanning tunneling microscope.
Dec 12th, 2011
Read more
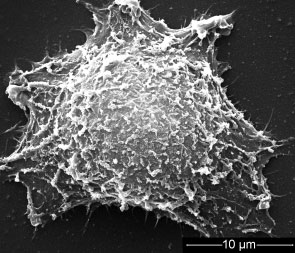 New pretreatment process delivers biocompatible, stable gold nanorods for tumor treatment.
New pretreatment process delivers biocompatible, stable gold nanorods for tumor treatment.
Dec 12th, 2011
Read more
Structural studies at Berkeley Lab's Advanced Light Source could point to reduced carbon emissions and stronger cements.
Dec 12th, 2011
Read more








 Subscribe to our Nanotechnology News feed
Subscribe to our Nanotechnology News feed