Showing Spotlights 953 - 960 of 2784 in category All (newest first):
 Nanoengineered functional textiles are going to revolutionize the clothing that you'll wear. The potential of nanotechnology in the development of new materials in the textile industry is considerable. This review discusses electronic and photonic nanotechnologies that are integrated with textiles and shows their applications in displays, sensing, and drug release within the context of performance, durability, and connectivity. On the one hand, existing functionality can be improved using nanotechnology and on the other, it could make possible the manufacture of textiles with entirely new properties or the combination of different functions in one textile material.
Nanoengineered functional textiles are going to revolutionize the clothing that you'll wear. The potential of nanotechnology in the development of new materials in the textile industry is considerable. This review discusses electronic and photonic nanotechnologies that are integrated with textiles and shows their applications in displays, sensing, and drug release within the context of performance, durability, and connectivity. On the one hand, existing functionality can be improved using nanotechnology and on the other, it could make possible the manufacture of textiles with entirely new properties or the combination of different functions in one textile material.
Feb 29th, 2016
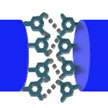 Not all electrocapacitive materials are intrinsically stretchable and various modified structures and electron/ion-inactive stretchable substrates have been utilized to introduce stretchability into conventionally rigid supercapacitors. Now, researchers have developed a multifunctional polyelectrolyte, achieving an electrochemically complete self-healability and 600% stretchability of supercapacitors. This work can be applied to other energy conversion and storage devices such as batteries, fuel cells, etc.
Not all electrocapacitive materials are intrinsically stretchable and various modified structures and electron/ion-inactive stretchable substrates have been utilized to introduce stretchability into conventionally rigid supercapacitors. Now, researchers have developed a multifunctional polyelectrolyte, achieving an electrochemically complete self-healability and 600% stretchability of supercapacitors. This work can be applied to other energy conversion and storage devices such as batteries, fuel cells, etc.
Feb 22nd, 2016
 Researchers have explored common inexpensive materials to demonstrate their valuable and advantageous properties for artificial skin development. They demonstrate a scalable fabrication approach using off-the-shelf household items such as aluminum foil, scotch tapes, sticky-notes, napkins and sponges to build 'Paper Skin'. Paper Skin promises to be an affordable all-in-one flexibel sensing platform, applicable for applications such as health monitoring, 3D touchscreens, and human-machine interfaces, where sensing diversity, surface adaptability, and large-area mapping all are essential.
Researchers have explored common inexpensive materials to demonstrate their valuable and advantageous properties for artificial skin development. They demonstrate a scalable fabrication approach using off-the-shelf household items such as aluminum foil, scotch tapes, sticky-notes, napkins and sponges to build 'Paper Skin'. Paper Skin promises to be an affordable all-in-one flexibel sensing platform, applicable for applications such as health monitoring, 3D touchscreens, and human-machine interfaces, where sensing diversity, surface adaptability, and large-area mapping all are essential.
Feb 19th, 2016
 By replacing dye inks with optical nanostructures, researchers have demonstrated the use of inkjet technology to create colored interference layers with high accuracy without the need for high-temperature fixing. The key to this technology is the ability to control the inkjet printing process to form nanostructures with high accuracy. Whereas conventional inkjet printing occurs in the microrange, this new work demonstrates that optical structures can be printed with much greater accuracy, which in particular opens the prospects in developing controlled interference printing of interference color images.
By replacing dye inks with optical nanostructures, researchers have demonstrated the use of inkjet technology to create colored interference layers with high accuracy without the need for high-temperature fixing. The key to this technology is the ability to control the inkjet printing process to form nanostructures with high accuracy. Whereas conventional inkjet printing occurs in the microrange, this new work demonstrates that optical structures can be printed with much greater accuracy, which in particular opens the prospects in developing controlled interference printing of interference color images.
Feb 11th, 2016
 Many virus detection platforms, including conventional fluorescent label-based ones, have limitations because they are time-intensive and not easily compatible with point-of-care use without the existence of significant infrastructure and expert staff. Researchers have now developed a technique capable of specifically visualizing label-free single viruses in complex solutions in real-time. This approach eliminates virtually all sample preparation.
Many virus detection platforms, including conventional fluorescent label-based ones, have limitations because they are time-intensive and not easily compatible with point-of-care use without the existence of significant infrastructure and expert staff. Researchers have now developed a technique capable of specifically visualizing label-free single viruses in complex solutions in real-time. This approach eliminates virtually all sample preparation.
Feb 5th, 2016
 The scaling up of nanomaterials in the broader context of materials science and engineering is the topic of a Perspective article, where the authors construct a roadmap for assembling nanoscale building blocks into bulk nanostructured materials, and define some of the critical challenges and goals. Two-dimenisonal sheets are uniquely well-suited in this roadmap for constructing dense, bulk-sized samples with scalable material performance or interesting emergent properties. But no matter what structures are used, when nanostructures with better-than-bulk material performances are used in bulk form, it is critical that those extraordinary nanoscale properties can be scaled to the macroscopic level.
The scaling up of nanomaterials in the broader context of materials science and engineering is the topic of a Perspective article, where the authors construct a roadmap for assembling nanoscale building blocks into bulk nanostructured materials, and define some of the critical challenges and goals. Two-dimenisonal sheets are uniquely well-suited in this roadmap for constructing dense, bulk-sized samples with scalable material performance or interesting emergent properties. But no matter what structures are used, when nanostructures with better-than-bulk material performances are used in bulk form, it is critical that those extraordinary nanoscale properties can be scaled to the macroscopic level.
Feb 4th, 2016
 Resistive random access memory (RRAM) is envisioned as a next generation non-volatile memory because of the simple device geometry, ease of fabrication and operation. The necessity of high-density information storage and its relevance in neuromorphic circuitry has gained much attention and led to the development of multilevel resistive switching (MRS) for multiple memory states. In a recent study, researchers have defined a new figure-of-merit to identify the efficiency of resistive switching devices with multiple memory states. This will assist researches as well as technologist in classifying and deciding the true merit of their memory devices.
Resistive random access memory (RRAM) is envisioned as a next generation non-volatile memory because of the simple device geometry, ease of fabrication and operation. The necessity of high-density information storage and its relevance in neuromorphic circuitry has gained much attention and led to the development of multilevel resistive switching (MRS) for multiple memory states. In a recent study, researchers have defined a new figure-of-merit to identify the efficiency of resistive switching devices with multiple memory states. This will assist researches as well as technologist in classifying and deciding the true merit of their memory devices.
Feb 1st, 2016
 Hierarchical porous carbon/graphene (HPC/HPG) materials have been intensively investigated over the past decades. These materials are demonstrated as promising electrode materials for various systems, such as lithium-ion batteries, lithium-sulfur batteries, supercapacitors, and fuel cells, with a remarkable capacity, high efficiency, long stability, and excellent rate capability. Researchers have now proposed the employment of hierarchical porous calcium oxide (CaO) particles as effective catalytic template for the facile CVD growth of graphene.
Hierarchical porous carbon/graphene (HPC/HPG) materials have been intensively investigated over the past decades. These materials are demonstrated as promising electrode materials for various systems, such as lithium-ion batteries, lithium-sulfur batteries, supercapacitors, and fuel cells, with a remarkable capacity, high efficiency, long stability, and excellent rate capability. Researchers have now proposed the employment of hierarchical porous calcium oxide (CaO) particles as effective catalytic template for the facile CVD growth of graphene.
Jan 26th, 2016
 Nanoengineered functional textiles are going to revolutionize the clothing that you'll wear. The potential of nanotechnology in the development of new materials in the textile industry is considerable. This review discusses electronic and photonic nanotechnologies that are integrated with textiles and shows their applications in displays, sensing, and drug release within the context of performance, durability, and connectivity. On the one hand, existing functionality can be improved using nanotechnology and on the other, it could make possible the manufacture of textiles with entirely new properties or the combination of different functions in one textile material.
Nanoengineered functional textiles are going to revolutionize the clothing that you'll wear. The potential of nanotechnology in the development of new materials in the textile industry is considerable. This review discusses electronic and photonic nanotechnologies that are integrated with textiles and shows their applications in displays, sensing, and drug release within the context of performance, durability, and connectivity. On the one hand, existing functionality can be improved using nanotechnology and on the other, it could make possible the manufacture of textiles with entirely new properties or the combination of different functions in one textile material.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





