Showing Spotlights 1025 - 1032 of 2785 in category All (newest first):
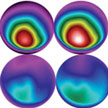 Microwave hyperthermia is one of the most important clinical thermotherapy techniques, however, it is often difficult to apply heat locally to a tumor. By bringing together experts in medicine with experts in nanotechnology, researchers aim to explore the possibilities of designing microwave sensitive agents, which focus the destructive effects of microwaves specifically onto tumor cells. Scientists have now proposed a novel approach to apply micro- and nanomaterials as microwave susceptible agents in tumor hyperthermia in vivo for the first time.
Microwave hyperthermia is one of the most important clinical thermotherapy techniques, however, it is often difficult to apply heat locally to a tumor. By bringing together experts in medicine with experts in nanotechnology, researchers aim to explore the possibilities of designing microwave sensitive agents, which focus the destructive effects of microwaves specifically onto tumor cells. Scientists have now proposed a novel approach to apply micro- and nanomaterials as microwave susceptible agents in tumor hyperthermia in vivo for the first time.
Jun 26th, 2015
 Observations made on a fern and an insect have led researchers to develop a nanofur structure that significantly reduces fluid drag. Both have surfaces covered by high density hairs which allow them to keep an air layer under water. This enables the bug to move nimbly and swiftly through the water by reducing the drag on its surface. Based on these observations, researchers have developed a very inexpensive, highly scalable method to produce a superhydrophobic, air retaining biomimetic surface - a 'nanofur' - which shows not only a high long-term stability but also a high resistance against additional applied pressure.
Observations made on a fern and an insect have led researchers to develop a nanofur structure that significantly reduces fluid drag. Both have surfaces covered by high density hairs which allow them to keep an air layer under water. This enables the bug to move nimbly and swiftly through the water by reducing the drag on its surface. Based on these observations, researchers have developed a very inexpensive, highly scalable method to produce a superhydrophobic, air retaining biomimetic surface - a 'nanofur' - which shows not only a high long-term stability but also a high resistance against additional applied pressure.
Jun 22nd, 2015
 Self-healing of a device is different from material self-healing because the devices contain electronic circuits. Self-healing of a device includes materials self-healing plus alignment of electrodes, which is very difficult but essential. Researchers have now come up with the idea of using magnetic force to assist alignment of electrodes in a circuit, facilitating self-healing of the whole device. To realize this idea, they designed and fabricated an electrically and mechanically self-healable yarn-based supercapacitor by wrapping magnetic electrodes with a self-healing carboxylated polyurethane shell.
Self-healing of a device is different from material self-healing because the devices contain electronic circuits. Self-healing of a device includes materials self-healing plus alignment of electrodes, which is very difficult but essential. Researchers have now come up with the idea of using magnetic force to assist alignment of electrodes in a circuit, facilitating self-healing of the whole device. To realize this idea, they designed and fabricated an electrically and mechanically self-healable yarn-based supercapacitor by wrapping magnetic electrodes with a self-healing carboxylated polyurethane shell.
Jun 19th, 2015
 In conventional nanosphere lithography, the nanosphere configurations in the layers are determined by a spontaneous self-assembly process. Therefore, the final configurations are limited to those with or close to the minimal free energy giving rise to very simple patterns. Researchers have now managed to circumvent this thermodynamical restriction by putting the monolayers in a confined environment and constructing the bilayers with sequential stacking, both of which are critical for the formation of moire patterns.
In conventional nanosphere lithography, the nanosphere configurations in the layers are determined by a spontaneous self-assembly process. Therefore, the final configurations are limited to those with or close to the minimal free energy giving rise to very simple patterns. Researchers have now managed to circumvent this thermodynamical restriction by putting the monolayers in a confined environment and constructing the bilayers with sequential stacking, both of which are critical for the formation of moire patterns.
Jun 18th, 2015
 While exploring the possibility to realize graphene-like nanostructures of boron, carbon's neighbor in the periodic table, a team of chemical engineers has discovered an entirely new family of 2-D compounds. They demonstrated exfoliation of a well-known superconductor magnesium diboride, a layered material that consists Mg atoms sandwiched in between born honeycomb planes. These nanosheets can be an order of magnitude more transparent compared to their cousin graphene.
While exploring the possibility to realize graphene-like nanostructures of boron, carbon's neighbor in the periodic table, a team of chemical engineers has discovered an entirely new family of 2-D compounds. They demonstrated exfoliation of a well-known superconductor magnesium diboride, a layered material that consists Mg atoms sandwiched in between born honeycomb planes. These nanosheets can be an order of magnitude more transparent compared to their cousin graphene.
Jun 17th, 2015
 The key challenges associated with the development of high performance MEMS and NEMS resonators for RF wireless communication and sensing applications are the isolation of energy-dissipating mechanisms and scaling of the device volume in the nanoscale size-range. Researchers show that graphene-electrode based piezoelectric NEMS resonators operate at their theoretical 'unloaded' frequency-limits, with significantly improved electromechanical performance compared to metal-electrode counterparts, despite their reduced volumes.
The key challenges associated with the development of high performance MEMS and NEMS resonators for RF wireless communication and sensing applications are the isolation of energy-dissipating mechanisms and scaling of the device volume in the nanoscale size-range. Researchers show that graphene-electrode based piezoelectric NEMS resonators operate at their theoretical 'unloaded' frequency-limits, with significantly improved electromechanical performance compared to metal-electrode counterparts, despite their reduced volumes.
Jun 16th, 2015
 Researchers have developed a novel 3D-printing based method to produce highly monodisperse core/shell capsules that can be loaded with biomolecules such as therapeutic drugs. The method provides a robust control over particle properties, passive release kinetics, and particle distributions throughout a 3D matrix. Furthermore, these capsules are rendered stimuli-responsive by incorporating gold nanorods into the polymer shell, allowing for highly selective photothermal rupture and triggered temporal release of the biomolecular payload.
Researchers have developed a novel 3D-printing based method to produce highly monodisperse core/shell capsules that can be loaded with biomolecules such as therapeutic drugs. The method provides a robust control over particle properties, passive release kinetics, and particle distributions throughout a 3D matrix. Furthermore, these capsules are rendered stimuli-responsive by incorporating gold nanorods into the polymer shell, allowing for highly selective photothermal rupture and triggered temporal release of the biomolecular payload.
Jun 15th, 2015
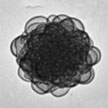 Over the last twenty years, scientists have developed many techniques to synthesize polymeric nanoparticles for a wide range of applications including surface coating, sensor technology, catalysis, and nanomedicine. However, the precise control of the size and shape of polymer nanoparticles remains challenging, and RDRP techniques still fall well short of producing large, well-defined macromolecules with the same size and degree of precision as nature (proteins, nucleic acids, etc.). In new work, researchers have developed a new technique to precisely control the size and shape of polymeric nanoparticles.
Over the last twenty years, scientists have developed many techniques to synthesize polymeric nanoparticles for a wide range of applications including surface coating, sensor technology, catalysis, and nanomedicine. However, the precise control of the size and shape of polymer nanoparticles remains challenging, and RDRP techniques still fall well short of producing large, well-defined macromolecules with the same size and degree of precision as nature (proteins, nucleic acids, etc.). In new work, researchers have developed a new technique to precisely control the size and shape of polymeric nanoparticles.
Jun 5th, 2015
 Microwave hyperthermia is one of the most important clinical thermotherapy techniques, however, it is often difficult to apply heat locally to a tumor. By bringing together experts in medicine with experts in nanotechnology, researchers aim to explore the possibilities of designing microwave sensitive agents, which focus the destructive effects of microwaves specifically onto tumor cells. Scientists have now proposed a novel approach to apply micro- and nanomaterials as microwave susceptible agents in tumor hyperthermia in vivo for the first time.
Microwave hyperthermia is one of the most important clinical thermotherapy techniques, however, it is often difficult to apply heat locally to a tumor. By bringing together experts in medicine with experts in nanotechnology, researchers aim to explore the possibilities of designing microwave sensitive agents, which focus the destructive effects of microwaves specifically onto tumor cells. Scientists have now proposed a novel approach to apply micro- and nanomaterials as microwave susceptible agents in tumor hyperthermia in vivo for the first time.

 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





