Showing Spotlights 1041 - 1048 of 2785 in category All (newest first):
 Upconversion luminesce materials are promising for widespread application ranging from optical devices to biodetection and cancer therapy, the near-infrared light excited upconversion materials are attracting much research attention. Researchers have achieved an ultra-strong and ultra-pure red upconversion in erbium and ytterbium co-doped lutetium oxyfluorides through size and morphology control. These nanoparticles, with extremely strong and pure upconversion are promising candidates as novel luminescent reagents for high-contrast bio-imaging and bio-labeling.
Upconversion luminesce materials are promising for widespread application ranging from optical devices to biodetection and cancer therapy, the near-infrared light excited upconversion materials are attracting much research attention. Researchers have achieved an ultra-strong and ultra-pure red upconversion in erbium and ytterbium co-doped lutetium oxyfluorides through size and morphology control. These nanoparticles, with extremely strong and pure upconversion are promising candidates as novel luminescent reagents for high-contrast bio-imaging and bio-labeling.
May 4th, 2015
 From a 3D printing perspective, graphene has been previously incorporated into 3D printed materials, but most of these constructs comprise no greater than about 20 volume % of the total solid of the composite, resulting in electrical properties that are significantly less than what has been achieced in new work. Here, researchers show that high volume fraction graphene composite constructs can be formed from an easily extrudable liquid ink into multi-centimeter scaled objects.
From a 3D printing perspective, graphene has been previously incorporated into 3D printed materials, but most of these constructs comprise no greater than about 20 volume % of the total solid of the composite, resulting in electrical properties that are significantly less than what has been achieced in new work. Here, researchers show that high volume fraction graphene composite constructs can be formed from an easily extrudable liquid ink into multi-centimeter scaled objects.
Apr 28th, 2015
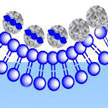 Biomineralization is the formation of inorganic materials in a biological environment, as it is found in bones, teeth and shells. Certain biominerals are also often associated with pathogeneses of tissues. The correlation of the composition of biominerals with pathogeneses of tissues has not been investigated systematically. In new work, researchers examine how the composition of biominerals correlates with the production of inflammatory cytokines associated with the stimulation of intracellular DNA sensors and by biominerals themselves.
Biomineralization is the formation of inorganic materials in a biological environment, as it is found in bones, teeth and shells. Certain biominerals are also often associated with pathogeneses of tissues. The correlation of the composition of biominerals with pathogeneses of tissues has not been investigated systematically. In new work, researchers examine how the composition of biominerals correlates with the production of inflammatory cytokines associated with the stimulation of intracellular DNA sensors and by biominerals themselves.
Apr 22nd, 2015
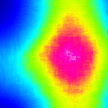 Plasmon lasers are promising nanoscale coherent sources of optical fields because they support ultra-small sizes and show ultra-fast dynamics. They can make possible single-molecule biodetectors, photonic circuits and high-speed optical communication systems. In new work, researchers have found a way to integrate liquid gain materials with gold nanoparticle arrays to achieve nanoscale plasmon lasing that can be tuned dynamical, reversibly, and in real time.
Plasmon lasers are promising nanoscale coherent sources of optical fields because they support ultra-small sizes and show ultra-fast dynamics. They can make possible single-molecule biodetectors, photonic circuits and high-speed optical communication systems. In new work, researchers have found a way to integrate liquid gain materials with gold nanoparticle arrays to achieve nanoscale plasmon lasing that can be tuned dynamical, reversibly, and in real time.
Apr 21st, 2015
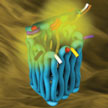 Research related to molecular logic gates is a fast growing and very active area and molecular devices have become the new frontier in computing. Researchers now have designed and synthesized self-assembled DNA complexes that sense two environmental signals and produce a fluorescent outputs corresponding to the operation of all six Boolean logic gates AND, NAND, OR, NOR, XOR, and XNOR. This study could help improvements in the fields of molecular computation and intelligent drug delivery.
Research related to molecular logic gates is a fast growing and very active area and molecular devices have become the new frontier in computing. Researchers now have designed and synthesized self-assembled DNA complexes that sense two environmental signals and produce a fluorescent outputs corresponding to the operation of all six Boolean logic gates AND, NAND, OR, NOR, XOR, and XNOR. This study could help improvements in the fields of molecular computation and intelligent drug delivery.
Apr 20th, 2015
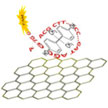 While the actual toxicity of Bisphenol A (BPA) is still debated, the direct measurement of BPA is difficult because of the weak response given by conventional electrochemical sensors, and current optical analysis methods are susceptible to the influence of interfering substances. A novel aptamer/graphene oxide FRET biosensor now provides a method for the rapid detection and risk assessment of BPA with high sensitivity and selectivity.
While the actual toxicity of Bisphenol A (BPA) is still debated, the direct measurement of BPA is difficult because of the weak response given by conventional electrochemical sensors, and current optical analysis methods are susceptible to the influence of interfering substances. A novel aptamer/graphene oxide FRET biosensor now provides a method for the rapid detection and risk assessment of BPA with high sensitivity and selectivity.
Apr 13th, 2015
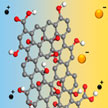 The commonly used separators in battery systems are porous polymer membranes, which separate the two electrodes while having little impact on the transportation of ions through the membrane. Polysulfides generated in a lithium-sulfur system can also diffuse freely through the membranes and react with a metal lithium anode, which results in the degradation of the battery's performance. If a novel, ion-selective but highly permeable separator can be developed, the shuttling of polysulfides and self-discharge would be effectively prevented, and both the energy density and power density of lithium-sulfur batteries could be significantly improved.
The commonly used separators in battery systems are porous polymer membranes, which separate the two electrodes while having little impact on the transportation of ions through the membrane. Polysulfides generated in a lithium-sulfur system can also diffuse freely through the membranes and react with a metal lithium anode, which results in the degradation of the battery's performance. If a novel, ion-selective but highly permeable separator can be developed, the shuttling of polysulfides and self-discharge would be effectively prevented, and both the energy density and power density of lithium-sulfur batteries could be significantly improved.
Mar 30th, 2015
 Novel materials designed and fabricated with the help of nanotechnologies offer the promise of radical technological development. Many of these will improve our quality of life, and develop our economies, but all will be measured against the overarching principle that we do not make some error, and harm ourselves and our environment by exposure to new forms of hazard. A publication explores recent developments in nanomaterials research, and possibilities for safe, practical and resource-efficient applications.
Novel materials designed and fabricated with the help of nanotechnologies offer the promise of radical technological development. Many of these will improve our quality of life, and develop our economies, but all will be measured against the overarching principle that we do not make some error, and harm ourselves and our environment by exposure to new forms of hazard. A publication explores recent developments in nanomaterials research, and possibilities for safe, practical and resource-efficient applications.
Mar 27th, 2015
 Upconversion luminesce materials are promising for widespread application ranging from optical devices to biodetection and cancer therapy, the near-infrared light excited upconversion materials are attracting much research attention. Researchers have achieved an ultra-strong and ultra-pure red upconversion in erbium and ytterbium co-doped lutetium oxyfluorides through size and morphology control. These nanoparticles, with extremely strong and pure upconversion are promising candidates as novel luminescent reagents for high-contrast bio-imaging and bio-labeling.
Upconversion luminesce materials are promising for widespread application ranging from optical devices to biodetection and cancer therapy, the near-infrared light excited upconversion materials are attracting much research attention. Researchers have achieved an ultra-strong and ultra-pure red upconversion in erbium and ytterbium co-doped lutetium oxyfluorides through size and morphology control. These nanoparticles, with extremely strong and pure upconversion are promising candidates as novel luminescent reagents for high-contrast bio-imaging and bio-labeling.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





