Showing Spotlights 1329 - 1336 of 2780 in category All (newest first):
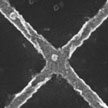 Ferromagnetic materials exhibit the so-called anomalous Hall effect (AHE), whereby the electrons flowing through the material experience a lateral force pushing them to one side as a result of the material's intrinsic magnetization. Although the AHE has been used in the field on nanotechnology to measure the magnetic behavior of nanoparticles (with sizes larger than 50 nm), nobody so far had tried to separate the signals of the individual particles. Researchers in Germany have now developed a simple technique which allows to measure the magnetic response of single ferromagnetic nanoparticles down to a radius of about 3.3 nm.
Ferromagnetic materials exhibit the so-called anomalous Hall effect (AHE), whereby the electrons flowing through the material experience a lateral force pushing them to one side as a result of the material's intrinsic magnetization. Although the AHE has been used in the field on nanotechnology to measure the magnetic behavior of nanoparticles (with sizes larger than 50 nm), nobody so far had tried to separate the signals of the individual particles. Researchers in Germany have now developed a simple technique which allows to measure the magnetic response of single ferromagnetic nanoparticles down to a radius of about 3.3 nm.
May 14th, 2013
 The low-frequency 1/f noise is a ubiquitous phenomenon found everywhere from fluctuations of human heart rates to fluctuations of electrical currents in semiconductor devices. An acceptable level of flicker 1/f noise is one of the key metrics that each new material has to pass before it can be used for practical devices. Graphene has shown a great potential for applications in high-frequency communications, analog circuits and sensors. The envisioned applications require a low level of 1/f noise, which contributes to the phase-noise of communication systems and limits the sensor sensitivity. Now, researchers have discovered a unique feature of 1/f noise in graphene, which can help understand its microscopic origin and develop new techniques for noise reduction.
The low-frequency 1/f noise is a ubiquitous phenomenon found everywhere from fluctuations of human heart rates to fluctuations of electrical currents in semiconductor devices. An acceptable level of flicker 1/f noise is one of the key metrics that each new material has to pass before it can be used for practical devices. Graphene has shown a great potential for applications in high-frequency communications, analog circuits and sensors. The envisioned applications require a low level of 1/f noise, which contributes to the phase-noise of communication systems and limits the sensor sensitivity. Now, researchers have discovered a unique feature of 1/f noise in graphene, which can help understand its microscopic origin and develop new techniques for noise reduction.
May 10th, 2013
 Nanopores are an exciting class of single molecule nanosensors. For several years now, nanopore technology has been developed as a biosensor at the single-molecule resolution to detect an array of biomedical molecules, such as DNA, RNA, protein, biotoxin, and various nanopore projects have been funded to develop the next generation of DNA sequencing technology. The sensing principle is based on the resistive pulse technique - molecules are detected as they pass through a single nanopore since during translocation the molecules exclude ions and therefore modulate the current. In new work, researchers have demonstrated the single molecule detection of a wide range of proteins with solid state glass nanopores.
Nanopores are an exciting class of single molecule nanosensors. For several years now, nanopore technology has been developed as a biosensor at the single-molecule resolution to detect an array of biomedical molecules, such as DNA, RNA, protein, biotoxin, and various nanopore projects have been funded to develop the next generation of DNA sequencing technology. The sensing principle is based on the resistive pulse technique - molecules are detected as they pass through a single nanopore since during translocation the molecules exclude ions and therefore modulate the current. In new work, researchers have demonstrated the single molecule detection of a wide range of proteins with solid state glass nanopores.
May 9th, 2013
 For a transistor to work properly, it must contain impurity atoms - called dopants - replacing the silicon atoms at certain places in the device. Given that modern transistor are approaching the atomic scale, the exact location of a single dopant atom becomes critical in determining the device functionality. In a different context, single dopant atoms in semiconductors have now proved to be an excellent platform to encode quantum information. Therefore, the exact location of single dopant atoms is also crucial to future quantum computers based on silicon. A new technique allows the accurate location of a single dopant atom in a nanoscale device, after the device has been fabricated, and without damaging or altering any of its functionalities.
For a transistor to work properly, it must contain impurity atoms - called dopants - replacing the silicon atoms at certain places in the device. Given that modern transistor are approaching the atomic scale, the exact location of a single dopant atom becomes critical in determining the device functionality. In a different context, single dopant atoms in semiconductors have now proved to be an excellent platform to encode quantum information. Therefore, the exact location of single dopant atoms is also crucial to future quantum computers based on silicon. A new technique allows the accurate location of a single dopant atom in a nanoscale device, after the device has been fabricated, and without damaging or altering any of its functionalities.
May 8th, 2013
 Every year, large quantities of antibiotics are released into lakes and rivers as a byproduct of their use in farming and medicine. Antibiotics in the environment can select for antibiotic-resistant bacterial populations, which can cause severe infections if they come into contact with humans or animals. In addition, antibiotics can disrupt the natural bacterial flora that plays a vital role in maintaining the balance of ecosystems. Scientists have now found that solar-powered proteoliposomes derived from bacteria can extract and store contaminants released into natural bodies of water,
Every year, large quantities of antibiotics are released into lakes and rivers as a byproduct of their use in farming and medicine. Antibiotics in the environment can select for antibiotic-resistant bacterial populations, which can cause severe infections if they come into contact with humans or animals. In addition, antibiotics can disrupt the natural bacterial flora that plays a vital role in maintaining the balance of ecosystems. Scientists have now found that solar-powered proteoliposomes derived from bacteria can extract and store contaminants released into natural bodies of water,
May 7th, 2013
 Most of the research efforts on developing synthesis methods for graphene has focused on flat substrates. However, direct growth of graphene layers on prepatterned substrates has remained elusive. In new work, resarchers have grown graphene in prepatterned copper-coated substrates, and they apply this protocol for the fabrication of MEMS devices, in particular, atomic force microscope probes. This layer of graphene improves the functionality of the probes by making them conductive and more resistant to wear.
Most of the research efforts on developing synthesis methods for graphene has focused on flat substrates. However, direct growth of graphene layers on prepatterned substrates has remained elusive. In new work, resarchers have grown graphene in prepatterned copper-coated substrates, and they apply this protocol for the fabrication of MEMS devices, in particular, atomic force microscope probes. This layer of graphene improves the functionality of the probes by making them conductive and more resistant to wear.
May 6th, 2013
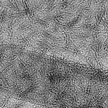 Integration of graphene and its functional derivatives into three-dimensional macroscopic structures is an essential step to exploit the advanced properties of graphene sheets for practical applications, such as chemical filters and electrodes for energy storage devices. In new work, researchers in Singapore report a facile, scalable, and solution-processable strategy to synchronously reduce and assemble graphene oxide sheets on metal surface into large scale chemically converted graphene films under ambient conditions. Compared with other techniques currently used to prepare large-scale graphene film, this novel processing is low-temperature with scalable and high-throughput capability.
Integration of graphene and its functional derivatives into three-dimensional macroscopic structures is an essential step to exploit the advanced properties of graphene sheets for practical applications, such as chemical filters and electrodes for energy storage devices. In new work, researchers in Singapore report a facile, scalable, and solution-processable strategy to synchronously reduce and assemble graphene oxide sheets on metal surface into large scale chemically converted graphene films under ambient conditions. Compared with other techniques currently used to prepare large-scale graphene film, this novel processing is low-temperature with scalable and high-throughput capability.
Apr 30th, 2013
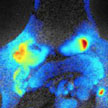 Scientists used a new type of biocompatible nanoparticle known as a 'porphysome' to detect and demarcate prostate tumors as small as 2mm in size. The tumors were imaged using a combination of positron emission tomography (PET) and optical imaging. PET allows doctors to detect, localize, and characterize prostate tumors non-invasively, while optical imaging allows them to find the edges of the tumor(s) during treatment. The porphysomes were also able to detect cancerous cells that had metastasized (spread) to the bone. This may allow doctors to find and treat cancer that has spread from the prostate to other parts of the body.
Scientists used a new type of biocompatible nanoparticle known as a 'porphysome' to detect and demarcate prostate tumors as small as 2mm in size. The tumors were imaged using a combination of positron emission tomography (PET) and optical imaging. PET allows doctors to detect, localize, and characterize prostate tumors non-invasively, while optical imaging allows them to find the edges of the tumor(s) during treatment. The porphysomes were also able to detect cancerous cells that had metastasized (spread) to the bone. This may allow doctors to find and treat cancer that has spread from the prostate to other parts of the body.
Apr 29th, 2013
 Ferromagnetic materials exhibit the so-called anomalous Hall effect (AHE), whereby the electrons flowing through the material experience a lateral force pushing them to one side as a result of the material's intrinsic magnetization. Although the AHE has been used in the field on nanotechnology to measure the magnetic behavior of nanoparticles (with sizes larger than 50 nm), nobody so far had tried to separate the signals of the individual particles. Researchers in Germany have now developed a simple technique which allows to measure the magnetic response of single ferromagnetic nanoparticles down to a radius of about 3.3 nm.
Ferromagnetic materials exhibit the so-called anomalous Hall effect (AHE), whereby the electrons flowing through the material experience a lateral force pushing them to one side as a result of the material's intrinsic magnetization. Although the AHE has been used in the field on nanotechnology to measure the magnetic behavior of nanoparticles (with sizes larger than 50 nm), nobody so far had tried to separate the signals of the individual particles. Researchers in Germany have now developed a simple technique which allows to measure the magnetic response of single ferromagnetic nanoparticles down to a radius of about 3.3 nm.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





