Showing Spotlights 129 - 136 of 544 in category All (newest first):
 Graphene bioelectronics has become a ground-breaking field that offers exciting opportunities for developing new kinds of sensors capable of establishing outstanding interfaces with soft tissue. Graphene-based transistors, as well as electrode arrays, have emerged as a special group of biosensors with their own peculiarities, advantages and drawbacks. A new review present sa comprehensive overview of the use of graphene for bioelectronics applications; specifically the focus is on interfacing graphene-based devices with electrogenic cells, such as cardiac and neuronal cells.
Graphene bioelectronics has become a ground-breaking field that offers exciting opportunities for developing new kinds of sensors capable of establishing outstanding interfaces with soft tissue. Graphene-based transistors, as well as electrode arrays, have emerged as a special group of biosensors with their own peculiarities, advantages and drawbacks. A new review present sa comprehensive overview of the use of graphene for bioelectronics applications; specifically the focus is on interfacing graphene-based devices with electrogenic cells, such as cardiac and neuronal cells.
Sep 3rd, 2018
 By employing 3D scaffolds with both aligned nanofibers and aligned interconnected macrochannels, researchers have taken a nanotechnology-based tissue engineering approach to accelerate the regeneration and repair of damaged tissues at the wound site by directing cells and tissues to grow towards the target site. Their hope is that this leads to the development of affordable and functional biodegradable wound dressings for accelerated healing of chronic skin wounds by promoting regeneration of local tissues.
By employing 3D scaffolds with both aligned nanofibers and aligned interconnected macrochannels, researchers have taken a nanotechnology-based tissue engineering approach to accelerate the regeneration and repair of damaged tissues at the wound site by directing cells and tissues to grow towards the target site. Their hope is that this leads to the development of affordable and functional biodegradable wound dressings for accelerated healing of chronic skin wounds by promoting regeneration of local tissues.
Aug 16th, 2018
 Scientists propose a novel tumor treatment strategy for simultaneous ablating tumor and inhibiting infection by using specially designed biodegradable metal-organic frameworks (MOFs). The strategy of simultaneous eradicating tumor tissue and inhibiting infection can inspire the researchers with novel progressing technologies for tumor therapy. This novel strategy opens the door for developing the nanomaterials that are sensitive to microwave irradiation and inhibiting infection to achieve noninvasive combined treatment in clinical practice.
Scientists propose a novel tumor treatment strategy for simultaneous ablating tumor and inhibiting infection by using specially designed biodegradable metal-organic frameworks (MOFs). The strategy of simultaneous eradicating tumor tissue and inhibiting infection can inspire the researchers with novel progressing technologies for tumor therapy. This novel strategy opens the door for developing the nanomaterials that are sensitive to microwave irradiation and inhibiting infection to achieve noninvasive combined treatment in clinical practice.
Jul 23rd, 2018
 Research on micro- and nanomachines has advanced from the observation and understanding of basic motion behavior to the constantly improving capabilities of performing complex tasks. This ranges from interaction with bacteria to propulsion based on cells and in vivo medical applications. A review looks at the applications of micro- and nanomachines in living biosystems from two aspects: the interaction of micro/nanomachines with other microscopic organisms or biological units, and the efforts toward application of micro/nanomachines in human body.
Research on micro- and nanomachines has advanced from the observation and understanding of basic motion behavior to the constantly improving capabilities of performing complex tasks. This ranges from interaction with bacteria to propulsion based on cells and in vivo medical applications. A review looks at the applications of micro- and nanomachines in living biosystems from two aspects: the interaction of micro/nanomachines with other microscopic organisms or biological units, and the efforts toward application of micro/nanomachines in human body.
Jul 5th, 2018
 Programmable nucleic acid nanoparticle (NANP) technology is a relatively new field that has already given rise to a host of self-assembling nucleic acid nanoparticles that are increasingly viewed as promising biological materials for medical applications. Programmable self-assembling NANPs are amenable to chemical modifications, control over functionalization, and consistent batch-to-batch formulation. Researchers present the first detailed and systematic study that involved 25 most representative NANPs, originally designed by different groups, screened for the induction of 29 different cytokines.
Programmable nucleic acid nanoparticle (NANP) technology is a relatively new field that has already given rise to a host of self-assembling nucleic acid nanoparticles that are increasingly viewed as promising biological materials for medical applications. Programmable self-assembling NANPs are amenable to chemical modifications, control over functionalization, and consistent batch-to-batch formulation. Researchers present the first detailed and systematic study that involved 25 most representative NANPs, originally designed by different groups, screened for the induction of 29 different cytokines.
Jun 25th, 2018
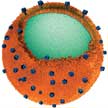 Cell membrane coating is a platform technology that presents a facile top-down method for designing nanocarriers with surfaces that directly replicate the highly complex functionalities necessary for effective biointerfacing. This new class of biomimetic nanoparticles combines the advantages of both natural and artificial nanomaterials. Cell membrane-coated nanoparticles are characterized by a synthetic nanoparticulate core cloaked by a layer of natural cell membrane.
Cell membrane coating is a platform technology that presents a facile top-down method for designing nanocarriers with surfaces that directly replicate the highly complex functionalities necessary for effective biointerfacing. This new class of biomimetic nanoparticles combines the advantages of both natural and artificial nanomaterials. Cell membrane-coated nanoparticles are characterized by a synthetic nanoparticulate core cloaked by a layer of natural cell membrane.
Jun 21st, 2018
 Drug abuse and dependence/addiction are complex disorders that are regulated by a wide range of interacting networks of genes and pathways that control a variety of phenotypes. Therefore, both identification of the at-risk population and treatment of the addiction disorders are strongly reliant on the development of new and innovative approaches for understanding the mechanisms underlying drug dependency and addiction. There are potential capacities of nanotechnology for the field of drug dependency and addiction.
Drug abuse and dependence/addiction are complex disorders that are regulated by a wide range of interacting networks of genes and pathways that control a variety of phenotypes. Therefore, both identification of the at-risk population and treatment of the addiction disorders are strongly reliant on the development of new and innovative approaches for understanding the mechanisms underlying drug dependency and addiction. There are potential capacities of nanotechnology for the field of drug dependency and addiction.
Jun 1st, 2018
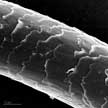 Scientists have discovered that the hierarchical micro- and nanostructures of human hair can be turned into hierarchical micro- and nanoparticles with a simple top-down procedure and be used as a novel type of biomaterial for medical applications. This strategy of preparing biomaterials from abundant human hair might provide a potent tool for producing autogenous materials from patients themselves to overcome the drawbacks of synthetic materials.
Scientists have discovered that the hierarchical micro- and nanostructures of human hair can be turned into hierarchical micro- and nanoparticles with a simple top-down procedure and be used as a novel type of biomaterial for medical applications. This strategy of preparing biomaterials from abundant human hair might provide a potent tool for producing autogenous materials from patients themselves to overcome the drawbacks of synthetic materials.
May 30th, 2018
 Graphene bioelectronics has become a ground-breaking field that offers exciting opportunities for developing new kinds of sensors capable of establishing outstanding interfaces with soft tissue. Graphene-based transistors, as well as electrode arrays, have emerged as a special group of biosensors with their own peculiarities, advantages and drawbacks. A new review present sa comprehensive overview of the use of graphene for bioelectronics applications; specifically the focus is on interfacing graphene-based devices with electrogenic cells, such as cardiac and neuronal cells.
Graphene bioelectronics has become a ground-breaking field that offers exciting opportunities for developing new kinds of sensors capable of establishing outstanding interfaces with soft tissue. Graphene-based transistors, as well as electrode arrays, have emerged as a special group of biosensors with their own peculiarities, advantages and drawbacks. A new review present sa comprehensive overview of the use of graphene for bioelectronics applications; specifically the focus is on interfacing graphene-based devices with electrogenic cells, such as cardiac and neuronal cells.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





