Showing Spotlights 129 - 136 of 315 in category All (newest first):
 Among various nanomaterial candidates for water treatment, metal oxides have been widely used as removal agents for various heavy metal ions and their removal capacity was found to be relatively reliable. The removal mechanism for heavy metal ions is thought to be the formation of a strong bond between metal ions and metal oxide surfaces. This strong complexation is advantageous for complete removal of heavy metal ions but it presents a drawback if one wants to design a reusable agent by reviving the reaction site for heavy metal ions. Precisely because the removal mechanism is based on the strong complex formation between metal ions and oxide surfaces, recycling of these removal agents has proved to be difficult. Offering a potential solution, researchers have demonstrated a recyclable removal agent for heavy metal ions by fabricating a core-in-shell structure based on a core of carbon nanotubes and an iron oxide microcapsule structure.
Among various nanomaterial candidates for water treatment, metal oxides have been widely used as removal agents for various heavy metal ions and their removal capacity was found to be relatively reliable. The removal mechanism for heavy metal ions is thought to be the formation of a strong bond between metal ions and metal oxide surfaces. This strong complexation is advantageous for complete removal of heavy metal ions but it presents a drawback if one wants to design a reusable agent by reviving the reaction site for heavy metal ions. Precisely because the removal mechanism is based on the strong complex formation between metal ions and oxide surfaces, recycling of these removal agents has proved to be difficult. Offering a potential solution, researchers have demonstrated a recyclable removal agent for heavy metal ions by fabricating a core-in-shell structure based on a core of carbon nanotubes and an iron oxide microcapsule structure.
Mar 3rd, 2010
 Materials engineers are keen to exploit the outstanding mechanical properties of carbon nanotubes for applications in fibers, composites, fabrics and other larger-scale structures and devices. The ability to fabricate continuous, multifunctional yarns represents an important step in this direction. The development of a continuous, weavable multilayered CNT yarn with superior mechanical, structural, surface, and electrical properties would open the way for a wide range of structural and functional applications, including composites, intelligent fabrics, catalyst supports, and sensors. Researchers in China now demonstrate the fabrication of a novel continuous yarn of CNTs with a multiple-layer structure by a CVD spinning process. The yarn consists of multiple monolayers of CNTs concentrically assembled in seamless tubules along the yarn axis.
Materials engineers are keen to exploit the outstanding mechanical properties of carbon nanotubes for applications in fibers, composites, fabrics and other larger-scale structures and devices. The ability to fabricate continuous, multifunctional yarns represents an important step in this direction. The development of a continuous, weavable multilayered CNT yarn with superior mechanical, structural, surface, and electrical properties would open the way for a wide range of structural and functional applications, including composites, intelligent fabrics, catalyst supports, and sensors. Researchers in China now demonstrate the fabrication of a novel continuous yarn of CNTs with a multiple-layer structure by a CVD spinning process. The yarn consists of multiple monolayers of CNTs concentrically assembled in seamless tubules along the yarn axis.
Feb 9th, 2010
 Carbon nanotubes are 'strange' nanostructures in a sense that they have both high mechanical strength and extreme flexibility. Deforming a carbon nanotube into any shape would not easily break the structure, and it recovers to original morphology in perfect manner. Researchers in China are exploiting this phenomenon by making CNT sponges consisting of a large amount of interconnected nanotubes, thus showing a combination of useful properties such as high porosity, super elasticity, robustness, and little weight. The nanotube sponges not only show exciting properties as a porous material but they also are very promising to be used practically in a short time. The production method is simple and scalable, the cost is low, and the sponges can find immediate use in many fields related to water purification.
Carbon nanotubes are 'strange' nanostructures in a sense that they have both high mechanical strength and extreme flexibility. Deforming a carbon nanotube into any shape would not easily break the structure, and it recovers to original morphology in perfect manner. Researchers in China are exploiting this phenomenon by making CNT sponges consisting of a large amount of interconnected nanotubes, thus showing a combination of useful properties such as high porosity, super elasticity, robustness, and little weight. The nanotube sponges not only show exciting properties as a porous material but they also are very promising to be used practically in a short time. The production method is simple and scalable, the cost is low, and the sponges can find immediate use in many fields related to water purification.
Feb 8th, 2010
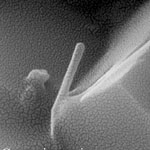 No other element in the periodic table bonds to itself in an extended network with the strength of the carbon-carbon bond. This special nature of carbon, combined with the molecular perfection of single-walled nanotubes endows these nanotubes with exceptional material properties, such as very high electrical and thermal conductivity, strength, stiffness, and toughness. As a result, single-walled carbon nanotubes (SWCNTs) are the strongest material known to science. SWCNTs potentially can add incredible strength, stiffness and electrical conductivity to all kinds of composite materials. Unfortunately, they are always held together in rope form due to their extremely small dimension and van der Waals attraction and their strength can neither be exploited nor measured. However, if several SWCNTs are concentrically nested in a confined space, the sliding issue can be resolved and the SWCNTs may share the applied tensile load to realize nanometer-scale materials remarkably stronger than the individual SWCNTs.
No other element in the periodic table bonds to itself in an extended network with the strength of the carbon-carbon bond. This special nature of carbon, combined with the molecular perfection of single-walled nanotubes endows these nanotubes with exceptional material properties, such as very high electrical and thermal conductivity, strength, stiffness, and toughness. As a result, single-walled carbon nanotubes (SWCNTs) are the strongest material known to science. SWCNTs potentially can add incredible strength, stiffness and electrical conductivity to all kinds of composite materials. Unfortunately, they are always held together in rope form due to their extremely small dimension and van der Waals attraction and their strength can neither be exploited nor measured. However, if several SWCNTs are concentrically nested in a confined space, the sliding issue can be resolved and the SWCNTs may share the applied tensile load to realize nanometer-scale materials remarkably stronger than the individual SWCNTs.
Dec 22nd, 2009
 Safe drinking water has been and increasingly will be a pressing issue for communities around the world. In developed countries it is about keeping water supplies safe while in the rest of the world it is about making it safe. The potential impact areas for nanotechnology in water applications are divided into three categories - treatment and remediation, sensing and detection, and pollution prevention. Within the category of sensing and detection, of particular interest is the development of new and enhanced sensors to detect biological and chemical contaminants at very low concentration levels. Testing of water against a spectrum of pathogens can potentially reduce the likelihood of many diseases from cancer to viral infections.
Safe drinking water has been and increasingly will be a pressing issue for communities around the world. In developed countries it is about keeping water supplies safe while in the rest of the world it is about making it safe. The potential impact areas for nanotechnology in water applications are divided into three categories - treatment and remediation, sensing and detection, and pollution prevention. Within the category of sensing and detection, of particular interest is the development of new and enhanced sensors to detect biological and chemical contaminants at very low concentration levels. Testing of water against a spectrum of pathogens can potentially reduce the likelihood of many diseases from cancer to viral infections.
Dec 8th, 2009
 A newly published antibacterial activity mechanism study demonstrates how a single walled carbon nanotube (SWCNT) kills bacteria by the physical puncture of bacterial membranes. The nanotubes would constantly attack the bacteria in solution, degrading the bacterial cell integrity and causing the cell death. This work elucidates several factors controlling the antibacterial activity of pristine SWCNTs and provides an insight in their toxicity mechanism. With regard to carbon nanotubes, in early toxicological studies, researchers obtained confounding results - in some studies nanotubes were toxic; in others, they were not. The apparent contradictions were actually a result of the materials that the researchers were using.
A newly published antibacterial activity mechanism study demonstrates how a single walled carbon nanotube (SWCNT) kills bacteria by the physical puncture of bacterial membranes. The nanotubes would constantly attack the bacteria in solution, degrading the bacterial cell integrity and causing the cell death. This work elucidates several factors controlling the antibacterial activity of pristine SWCNTs and provides an insight in their toxicity mechanism. With regard to carbon nanotubes, in early toxicological studies, researchers obtained confounding results - in some studies nanotubes were toxic; in others, they were not. The apparent contradictions were actually a result of the materials that the researchers were using.
Nov 10th, 2009
 New research reported this week has now established an industrially relevant process for assembling carbon nanotubes that allows them to efficiently be made into fibers, coatings and films - the basic forms of material that can be used in engineering applications. The most common of processing nanotubes into neat fibers - apart from 'dry' methods where they are spun directly into ropes and yarns - are 'wet' methods where CNTs are dispersed into a liquid and solution-spun into fiber. Currently, these processes yield fibers whose properties are not sufficiently close to optimal. Successful carbon nanotube assembly begins with control of dispersion and phase behavior and requires a scientific understanding of flow, colloidal interactions and solvent removal.
New research reported this week has now established an industrially relevant process for assembling carbon nanotubes that allows them to efficiently be made into fibers, coatings and films - the basic forms of material that can be used in engineering applications. The most common of processing nanotubes into neat fibers - apart from 'dry' methods where they are spun directly into ropes and yarns - are 'wet' methods where CNTs are dispersed into a liquid and solution-spun into fiber. Currently, these processes yield fibers whose properties are not sufficiently close to optimal. Successful carbon nanotube assembly begins with control of dispersion and phase behavior and requires a scientific understanding of flow, colloidal interactions and solvent removal.
Nov 5th, 2009
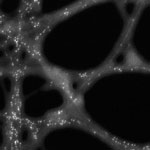 Due to their unique structural and electrical properties, carbon nanotubes have been extensively investigated as promising catalyst supports to improve the efficiency of direct ethanol/methanol fuel cells. CNTs have a significantly higher electronic conductivity and an extremely higher specific surface area in comparison with the most widely-used Vulcan XC-72R carbon support. Several approaches, such as electrochemical reduction, electroless deposition, spontaneous reduction, sonochemical technique, microwave-heated polyol process, and nanoparticle decoration on chemically oxidized nanotube sidewalls, have been reported to form CNT-supported platinum catalysts. Some remarkable progress has been made in synthesis techniques; however, pioneering breakthroughs have not been made yet in terms of cost-effectiveness catalyst activity, durability, and chemical-electrochemical stability. Nanotechnology researchers in the U.S. have now discovered that platinum nanoparticles selectively grow on carbon nanotubes in accordance with single-stranded DNA locations.
Due to their unique structural and electrical properties, carbon nanotubes have been extensively investigated as promising catalyst supports to improve the efficiency of direct ethanol/methanol fuel cells. CNTs have a significantly higher electronic conductivity and an extremely higher specific surface area in comparison with the most widely-used Vulcan XC-72R carbon support. Several approaches, such as electrochemical reduction, electroless deposition, spontaneous reduction, sonochemical technique, microwave-heated polyol process, and nanoparticle decoration on chemically oxidized nanotube sidewalls, have been reported to form CNT-supported platinum catalysts. Some remarkable progress has been made in synthesis techniques; however, pioneering breakthroughs have not been made yet in terms of cost-effectiveness catalyst activity, durability, and chemical-electrochemical stability. Nanotechnology researchers in the U.S. have now discovered that platinum nanoparticles selectively grow on carbon nanotubes in accordance with single-stranded DNA locations.
Oct 28th, 2009
 Among various nanomaterial candidates for water treatment, metal oxides have been widely used as removal agents for various heavy metal ions and their removal capacity was found to be relatively reliable. The removal mechanism for heavy metal ions is thought to be the formation of a strong bond between metal ions and metal oxide surfaces. This strong complexation is advantageous for complete removal of heavy metal ions but it presents a drawback if one wants to design a reusable agent by reviving the reaction site for heavy metal ions. Precisely because the removal mechanism is based on the strong complex formation between metal ions and oxide surfaces, recycling of these removal agents has proved to be difficult. Offering a potential solution, researchers have demonstrated a recyclable removal agent for heavy metal ions by fabricating a core-in-shell structure based on a core of carbon nanotubes and an iron oxide microcapsule structure.
Among various nanomaterial candidates for water treatment, metal oxides have been widely used as removal agents for various heavy metal ions and their removal capacity was found to be relatively reliable. The removal mechanism for heavy metal ions is thought to be the formation of a strong bond between metal ions and metal oxide surfaces. This strong complexation is advantageous for complete removal of heavy metal ions but it presents a drawback if one wants to design a reusable agent by reviving the reaction site for heavy metal ions. Precisely because the removal mechanism is based on the strong complex formation between metal ions and oxide surfaces, recycling of these removal agents has proved to be difficult. Offering a potential solution, researchers have demonstrated a recyclable removal agent for heavy metal ions by fabricating a core-in-shell structure based on a core of carbon nanotubes and an iron oxide microcapsule structure.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





