Showing Spotlights 1441 - 1448 of 2777 in category All (newest first):
 Nanoporous alumina membranes are used in a wide range of applications, from photonics and sensors to bioelectronics or filtration membranes, since they are basically a 'universal' mold for making zero- or one-dimensional nanostructures of mostly any material or compound. With current fabrication processes, the main limitations of porous alumina templates are their pore size, which cannot be smaller than 25nm, and their polydomain structure, which prevents the possibility of addressing each nanopore individually for electronics applications. A new nanofabrication process by researchers from France and Germany allows to reduce the pore diameter while maintaining the self-ordering and keeping the lattice constant. This led to a new family of AAO templates with identical pores with a diameter below 10nm and a porosity of 3.5%.
Nanoporous alumina membranes are used in a wide range of applications, from photonics and sensors to bioelectronics or filtration membranes, since they are basically a 'universal' mold for making zero- or one-dimensional nanostructures of mostly any material or compound. With current fabrication processes, the main limitations of porous alumina templates are their pore size, which cannot be smaller than 25nm, and their polydomain structure, which prevents the possibility of addressing each nanopore individually for electronics applications. A new nanofabrication process by researchers from France and Germany allows to reduce the pore diameter while maintaining the self-ordering and keeping the lattice constant. This led to a new family of AAO templates with identical pores with a diameter below 10nm and a porosity of 3.5%.
Aug 6th, 2012
 Nitrogen-doped carbon nanotubes (CNTs) have been extensively investigated for fuel cell applications due to their excellent electrocatalytic properties. However, their biomedical applications were comparatively less investigated despite reports of their better biocompatibility. When considering carbon nanotubes for drug delivery applications, it is desirable to develop strategies that allow utilize their hollow inner cavities for maximum loading capacity. Small size and facile surface modification are also preferable with regard to their biomedical compatibility. Nitrogen-doped CNTs have been already previously demonstrated to have better biocompatibility and mitigated cytotoxicity as compared to traditional undoped pristine CNTs. Taking advantage of this, researchers used nitrogen doping of CNTs which resulted in formation of cup-shaped compartments in CNTs uniquely suitable for encapsulation. The resulting nitrogen-doped carbon nanotube cups can be corked by gold nanoparticles to form enclosed nanocapsules.
Nitrogen-doped carbon nanotubes (CNTs) have been extensively investigated for fuel cell applications due to their excellent electrocatalytic properties. However, their biomedical applications were comparatively less investigated despite reports of their better biocompatibility. When considering carbon nanotubes for drug delivery applications, it is desirable to develop strategies that allow utilize their hollow inner cavities for maximum loading capacity. Small size and facile surface modification are also preferable with regard to their biomedical compatibility. Nitrogen-doped CNTs have been already previously demonstrated to have better biocompatibility and mitigated cytotoxicity as compared to traditional undoped pristine CNTs. Taking advantage of this, researchers used nitrogen doping of CNTs which resulted in formation of cup-shaped compartments in CNTs uniquely suitable for encapsulation. The resulting nitrogen-doped carbon nanotube cups can be corked by gold nanoparticles to form enclosed nanocapsules.
Aug 2nd, 2012
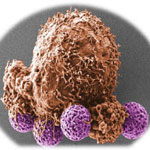 Early and accurate detection of cancer is critical for successful cancer therapies. In most cases, a tissue biopsy is the initial means of making a diagnosis. With increasing accuracy, 'liquid biopsies' - where circulating tumor cells (CTCs) are isolated from blood samples - are becoming a viable complement or even alternative to invasive biopsies of metastatic tumors. CTC is of great interest for evaluating cancer dissemination, predicting patient prognosis, and also for the evaluation of therapeutic treatments. In new work, researchers describe a rapid and simple electrochemical biosensing strategy to quantify circulating tumour cells based on the simultaneous use of antibody-coated magnetic beads, which selectively bind to the cancer cells for subsequent magnetic isolation, and antibody-coated gold nanoparticles, to also selectively bind to the cancer cells for final electrochemical detection.
Early and accurate detection of cancer is critical for successful cancer therapies. In most cases, a tissue biopsy is the initial means of making a diagnosis. With increasing accuracy, 'liquid biopsies' - where circulating tumor cells (CTCs) are isolated from blood samples - are becoming a viable complement or even alternative to invasive biopsies of metastatic tumors. CTC is of great interest for evaluating cancer dissemination, predicting patient prognosis, and also for the evaluation of therapeutic treatments. In new work, researchers describe a rapid and simple electrochemical biosensing strategy to quantify circulating tumour cells based on the simultaneous use of antibody-coated magnetic beads, which selectively bind to the cancer cells for subsequent magnetic isolation, and antibody-coated gold nanoparticles, to also selectively bind to the cancer cells for final electrochemical detection.
Aug 1st, 2012
 The emission of light by a single molecule is a cornerstone of nano-optics that will enable applications in quantum information processing or single-molecule spectroscopy. However, a key challenge in nano-optics is to bring light to and collect light from nano-scale systems. In conventional electronics, the interconnect between locally stored and radiated signals, for example radio broadcasts or mobile phone transmissions, is formed by antennas. For an antenna to work at the wavelength of light it is necessary to downscale the structure by the same factor as the wavelength or the frequency of the wave, i.e. roughly by a factor of 10 million. Once the nanofabrication issues are sorted out, nano-optical antennas could become ubiquitous in all applications based on light-matter interactions such as sensing, light emission (e.g. LEDs) and detection, as well as light harvesting, i.e. for solar cell applications.
The emission of light by a single molecule is a cornerstone of nano-optics that will enable applications in quantum information processing or single-molecule spectroscopy. However, a key challenge in nano-optics is to bring light to and collect light from nano-scale systems. In conventional electronics, the interconnect between locally stored and radiated signals, for example radio broadcasts or mobile phone transmissions, is formed by antennas. For an antenna to work at the wavelength of light it is necessary to downscale the structure by the same factor as the wavelength or the frequency of the wave, i.e. roughly by a factor of 10 million. Once the nanofabrication issues are sorted out, nano-optical antennas could become ubiquitous in all applications based on light-matter interactions such as sensing, light emission (e.g. LEDs) and detection, as well as light harvesting, i.e. for solar cell applications.
Jul 31st, 2012
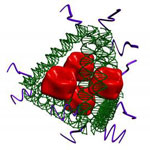 Vaccination is one of the most effective ways to prevent microbial infection. Synthetic vaccines can combine a portion of a microbe, known as an 'antigen' together with an adjuvant that stimulates the immune system. Delivering both the adjuvant and antigen to the appropriate immune cells is challenging. DNA nanotechnology may provide a solution by acting as a scaffold to co-deliver both antigen and adjuvant. However, the potential of DNA nanostructure-based vaccines has only been demonstrated in vitro. Now, a team of researchers based out of Arizona State University demonstrated that DNA nanostructures with appended adjuvants could elicit antibody production against a model antigen in mice.
Vaccination is one of the most effective ways to prevent microbial infection. Synthetic vaccines can combine a portion of a microbe, known as an 'antigen' together with an adjuvant that stimulates the immune system. Delivering both the adjuvant and antigen to the appropriate immune cells is challenging. DNA nanotechnology may provide a solution by acting as a scaffold to co-deliver both antigen and adjuvant. However, the potential of DNA nanostructure-based vaccines has only been demonstrated in vitro. Now, a team of researchers based out of Arizona State University demonstrated that DNA nanostructures with appended adjuvants could elicit antibody production against a model antigen in mice.
Jul 30th, 2012
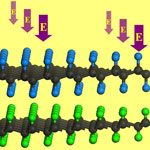 Researchers are putting great efforts into developing techniques to integrate graphene into nanoelectronic devices. Unfortunately, graphene has no band gap - a critical prerequisite for transistors - which essentially restricts its wider applications in nanoelectronics. Among the various techniques developed toward introducing a bandgap in graphene, hydrogenation or fluorination can efficiently solve this problem as they can open a considerable energy gap in the band structure of graphene. However, the experimentally realized fully hydrogenated and fluorinated graphene - namely graphane and fluorographene, respectively - both have a very large energy gap, which constrains their applications in electronics. Thus at present an urgent task is to find a feasible way which could reduce the energy gap of graphane or fluorographene into a desirable range. In new work, researchers have now demonstrated theoretically, using density functional theory computations, that graphane and fluorographene can be paired together through the C-H���F-C hydrogen bonds.
Researchers are putting great efforts into developing techniques to integrate graphene into nanoelectronic devices. Unfortunately, graphene has no band gap - a critical prerequisite for transistors - which essentially restricts its wider applications in nanoelectronics. Among the various techniques developed toward introducing a bandgap in graphene, hydrogenation or fluorination can efficiently solve this problem as they can open a considerable energy gap in the band structure of graphene. However, the experimentally realized fully hydrogenated and fluorinated graphene - namely graphane and fluorographene, respectively - both have a very large energy gap, which constrains their applications in electronics. Thus at present an urgent task is to find a feasible way which could reduce the energy gap of graphane or fluorographene into a desirable range. In new work, researchers have now demonstrated theoretically, using density functional theory computations, that graphane and fluorographene can be paired together through the C-H���F-C hydrogen bonds.
Jul 27th, 2012
 A major obstacle in today's chemical cancer therapies is to achieve specific drug accumulation at tumor sites and even tumor cells, because most chemotherapeutic agents are insoluble and instable, and can not differentiate between diseased and healthy cells. This often leads to severe adverse effects on healthy tissues and limits the maximum dose that can safely administered to patients. This issue becomes more serious in the case of some highly effective therapeutic agents, given that most potent anticancer drugs exhibit acute toxicity and narrow therapeutic window, and the clinical utility may not be possible without a powerful delivery systems equipped with smart properties that can allow them to precisely access the target tissues and cells. Targeted delivery approaches using nanomedicines will be able to provide solutions to these problems.
A major obstacle in today's chemical cancer therapies is to achieve specific drug accumulation at tumor sites and even tumor cells, because most chemotherapeutic agents are insoluble and instable, and can not differentiate between diseased and healthy cells. This often leads to severe adverse effects on healthy tissues and limits the maximum dose that can safely administered to patients. This issue becomes more serious in the case of some highly effective therapeutic agents, given that most potent anticancer drugs exhibit acute toxicity and narrow therapeutic window, and the clinical utility may not be possible without a powerful delivery systems equipped with smart properties that can allow them to precisely access the target tissues and cells. Targeted delivery approaches using nanomedicines will be able to provide solutions to these problems.
Jul 25th, 2012
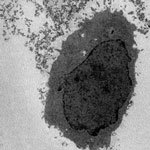 When a nanomaterial enters a physiological environment, it often adsorbs suspended biomolecules including proteins, lipids, small molecules, saccharides, and nucleic acids. These biomolecules define a new interface between the nanomaterial and its surroundings that mediates cellular association and response. In a recent article appearing in ACS Nano, Drs. Anna Salvati, Kenneth Dawson, and their colleagues at the University College in Dublin, Ireland, show that if nanoparticles are exposed directly to cells in the absence of suspended biomolecules, the nanoparticles will extract biomolecules from cells themselves. These findings suggest that in vitro toxicological or cell uptake studies performed in the absence of added serum may not be relevant in vivo.
When a nanomaterial enters a physiological environment, it often adsorbs suspended biomolecules including proteins, lipids, small molecules, saccharides, and nucleic acids. These biomolecules define a new interface between the nanomaterial and its surroundings that mediates cellular association and response. In a recent article appearing in ACS Nano, Drs. Anna Salvati, Kenneth Dawson, and their colleagues at the University College in Dublin, Ireland, show that if nanoparticles are exposed directly to cells in the absence of suspended biomolecules, the nanoparticles will extract biomolecules from cells themselves. These findings suggest that in vitro toxicological or cell uptake studies performed in the absence of added serum may not be relevant in vivo.
Jul 24th, 2012
 Nanoporous alumina membranes are used in a wide range of applications, from photonics and sensors to bioelectronics or filtration membranes, since they are basically a 'universal' mold for making zero- or one-dimensional nanostructures of mostly any material or compound. With current fabrication processes, the main limitations of porous alumina templates are their pore size, which cannot be smaller than 25nm, and their polydomain structure, which prevents the possibility of addressing each nanopore individually for electronics applications. A new nanofabrication process by researchers from France and Germany allows to reduce the pore diameter while maintaining the self-ordering and keeping the lattice constant. This led to a new family of AAO templates with identical pores with a diameter below 10nm and a porosity of 3.5%.
Nanoporous alumina membranes are used in a wide range of applications, from photonics and sensors to bioelectronics or filtration membranes, since they are basically a 'universal' mold for making zero- or one-dimensional nanostructures of mostly any material or compound. With current fabrication processes, the main limitations of porous alumina templates are their pore size, which cannot be smaller than 25nm, and their polydomain structure, which prevents the possibility of addressing each nanopore individually for electronics applications. A new nanofabrication process by researchers from France and Germany allows to reduce the pore diameter while maintaining the self-ordering and keeping the lattice constant. This led to a new family of AAO templates with identical pores with a diameter below 10nm and a porosity of 3.5%.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





