Showing Spotlights 1617 - 1624 of 2779 in category All (newest first):
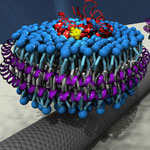 Integrating biological molecules or even complex molecular machines with man-made nanoelectronic devices is one of the ultimate goals of bionanotechnology. Already there is a growing community of researchers interested in this area of bio/nano integration where biological components are interfaced with inorganic nanomaterials to create new devices and systems that combine the desirable properties of each system. One particular nanomaterial used in this kind of research are carbon nanotubes (CNTs). Scientists now report the integration of a CNT transistor with olfactory receptor proteins. The ultimate goal of this type of research is to transfer the sensing properties of biological molecular systems to artificial electronic devices.
Integrating biological molecules or even complex molecular machines with man-made nanoelectronic devices is one of the ultimate goals of bionanotechnology. Already there is a growing community of researchers interested in this area of bio/nano integration where biological components are interfaced with inorganic nanomaterials to create new devices and systems that combine the desirable properties of each system. One particular nanomaterial used in this kind of research are carbon nanotubes (CNTs). Scientists now report the integration of a CNT transistor with olfactory receptor proteins. The ultimate goal of this type of research is to transfer the sensing properties of biological molecular systems to artificial electronic devices.
Jul 21st, 2011
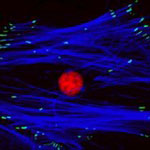 Currently, when adult stem cells are harvested from a patient, they are cultured in the laboratory to increase the initial yield of cells and create a batch of sufficient volume to kick-start the process of cellular regeneration when they are re-introduced back into the patient. The process of culturing is made more difficult by spontaneous stem cell differentiation, where stem cells grown on standard plastic tissue culture surfaces do not expand to create new stem cells but instead create other cells which are of no use in therapy. New findings show that nanoscale patterning is a powerful tool for the non-invasive manipulation of stem cells. Their facile fabrication process employed, a range of thermoplastics that can be processed with exquisite reproducibility down to 5 nm fidelity using injection moulding approaches, offers unique potential for the generation of cell culture platforms for the up-scale of autologous cells for clinical use.
Currently, when adult stem cells are harvested from a patient, they are cultured in the laboratory to increase the initial yield of cells and create a batch of sufficient volume to kick-start the process of cellular regeneration when they are re-introduced back into the patient. The process of culturing is made more difficult by spontaneous stem cell differentiation, where stem cells grown on standard plastic tissue culture surfaces do not expand to create new stem cells but instead create other cells which are of no use in therapy. New findings show that nanoscale patterning is a powerful tool for the non-invasive manipulation of stem cells. Their facile fabrication process employed, a range of thermoplastics that can be processed with exquisite reproducibility down to 5 nm fidelity using injection moulding approaches, offers unique potential for the generation of cell culture platforms for the up-scale of autologous cells for clinical use.
Jul 20th, 2011
 There is a physical and electrical disconnect between the world of electronics and the world of biology. Electronics tend to be rigid, operate using electrons, and are inherently two-dimensional. The brain, as a basis for comparison, is soft, operates using ions, and is three-dimensional. Researchers have therefore been looking to find different routes to create biocompatible devices that work well in wet environments like biological systems. In an exiting new development, researchers from North Carolina State University have fabricated a memory device that is soft, entirely based on liquid-based matter, and functions well in wet environments - opening the door to a new generation of biocompatible electronic devices.
There is a physical and electrical disconnect between the world of electronics and the world of biology. Electronics tend to be rigid, operate using electrons, and are inherently two-dimensional. The brain, as a basis for comparison, is soft, operates using ions, and is three-dimensional. Researchers have therefore been looking to find different routes to create biocompatible devices that work well in wet environments like biological systems. In an exiting new development, researchers from North Carolina State University have fabricated a memory device that is soft, entirely based on liquid-based matter, and functions well in wet environments - opening the door to a new generation of biocompatible electronic devices.
Jul 19th, 2011
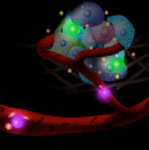 Monitoring cell functions and cell-to-cell communication has enormous implications for cell biology, regenerative medicine and tracking the fate of transplanted cells in cell therapy. Unfortunately, probing what cells 'see' and how they respond in real time to surrounding signals (i.e. cytokines) has been a major challenge. Now, a simple cell-surface sensor platform that permits signalling to be monitored within the cellular environment, in real time, in vitro and most likely also in vivo, can potentially address this problem. Researchers have developed a platform technology where cell-surface immobilized nanosensors allow them to monitor the cellular nano environment and cell-cell communication in real-time, at a single cell level and with potential unprecedented spatial and temporal resolution.
Monitoring cell functions and cell-to-cell communication has enormous implications for cell biology, regenerative medicine and tracking the fate of transplanted cells in cell therapy. Unfortunately, probing what cells 'see' and how they respond in real time to surrounding signals (i.e. cytokines) has been a major challenge. Now, a simple cell-surface sensor platform that permits signalling to be monitored within the cellular environment, in real time, in vitro and most likely also in vivo, can potentially address this problem. Researchers have developed a platform technology where cell-surface immobilized nanosensors allow them to monitor the cellular nano environment and cell-cell communication in real-time, at a single cell level and with potential unprecedented spatial and temporal resolution.
Jul 18th, 2011
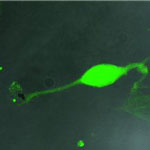 There is a growing body of research on using carbon nanotubes (CNTs) and other nanomaterials in neural engineering. Scientists are already exploring the feasibility of using CNTs to probe neural activity. With this research comes the need to develop a unified approach when assessing the toxicity of CNT in neurons. However, a complex picture emerges from the reported data: is it feasible to develop CNT-based devices as drug delivery vectors? Ultimately, are soluble CNT neurotoxic, and, if yes, to what degree? Given the often conflicting results of research reports on the biocompatibility of soluble CNT when administered to neurons in the central nervous system, a review article helps to clarify which aspects (technical or methodological) of these studies may be responsible for their heterogeneous conclusions.
There is a growing body of research on using carbon nanotubes (CNTs) and other nanomaterials in neural engineering. Scientists are already exploring the feasibility of using CNTs to probe neural activity. With this research comes the need to develop a unified approach when assessing the toxicity of CNT in neurons. However, a complex picture emerges from the reported data: is it feasible to develop CNT-based devices as drug delivery vectors? Ultimately, are soluble CNT neurotoxic, and, if yes, to what degree? Given the often conflicting results of research reports on the biocompatibility of soluble CNT when administered to neurons in the central nervous system, a review article helps to clarify which aspects (technical or methodological) of these studies may be responsible for their heterogeneous conclusions.
Jul 14th, 2011
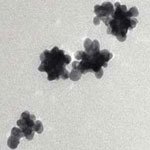 For more than 5000 years, noble metals have been known to be impervious to magnetic fields. Significant scientific breakthroughs in the recent past have led to making either 13 atoms clusters of platinum or organic molecule capped noble metal nanoparticles magnetic. By carefully controlling the size, shape, and composition of noble metals - especially in confined nanoscale systems - their properties can be modified, potentially leading to significantly advanced nanomaterial-based applications such as catalysis, self-assembly, novel metamaterials, improved MRI research, and spintronics. Now, researchers have developed simple method to make nano- and macro-particles of noble metals magnetic.
For more than 5000 years, noble metals have been known to be impervious to magnetic fields. Significant scientific breakthroughs in the recent past have led to making either 13 atoms clusters of platinum or organic molecule capped noble metal nanoparticles magnetic. By carefully controlling the size, shape, and composition of noble metals - especially in confined nanoscale systems - their properties can be modified, potentially leading to significantly advanced nanomaterial-based applications such as catalysis, self-assembly, novel metamaterials, improved MRI research, and spintronics. Now, researchers have developed simple method to make nano- and macro-particles of noble metals magnetic.
Jul 12th, 2011
 Energy generation and storage is an important issue at the nanoscale. For tiny devices such as nano- and micro-electromechanical systems, autonomous power sources are crucial for practical applications. Progress is being made in designing and fabricating nanoscale power generators. But, as with the large, macroscale systems of future smart grids, there might be times when powered nanodevices need to bridge a slump in power generation/harvesting or they might be designed to run on stored energy altogether for a limited period of time. Researchers in China are now proposing that the high energy density and power density of carbon nanotubes makes them promising materials for the storage of mechanical energy. The team provides a structural model towards mechanical energy storage for nanodevices and also demonstrates a method to characterize and manipulate ultralong CNTs.
Energy generation and storage is an important issue at the nanoscale. For tiny devices such as nano- and micro-electromechanical systems, autonomous power sources are crucial for practical applications. Progress is being made in designing and fabricating nanoscale power generators. But, as with the large, macroscale systems of future smart grids, there might be times when powered nanodevices need to bridge a slump in power generation/harvesting or they might be designed to run on stored energy altogether for a limited period of time. Researchers in China are now proposing that the high energy density and power density of carbon nanotubes makes them promising materials for the storage of mechanical energy. The team provides a structural model towards mechanical energy storage for nanodevices and also demonstrates a method to characterize and manipulate ultralong CNTs.
Jul 11th, 2011
 Silicone elastomers are widely used for biomedical applications and products. One major challenge for biomedical applications is to control the ingrowth of silicone-based implants and to avoid bacterial infections on device surfaces. The use of ions from metals like silver and copper is a promising, long-lasting method to achieve such bioactive effects. Researchers have now found a novel effect caused by a combination of copper and silver nanoparticles in silicone. By fabricating bioactive nanocomposite materials that release these ions in specific concentration levels and during a long time, manufacturers can control the bioactive effects of their medical devices or implants.
Silicone elastomers are widely used for biomedical applications and products. One major challenge for biomedical applications is to control the ingrowth of silicone-based implants and to avoid bacterial infections on device surfaces. The use of ions from metals like silver and copper is a promising, long-lasting method to achieve such bioactive effects. Researchers have now found a novel effect caused by a combination of copper and silver nanoparticles in silicone. By fabricating bioactive nanocomposite materials that release these ions in specific concentration levels and during a long time, manufacturers can control the bioactive effects of their medical devices or implants.
Jul 8th, 2011
 Integrating biological molecules or even complex molecular machines with man-made nanoelectronic devices is one of the ultimate goals of bionanotechnology. Already there is a growing community of researchers interested in this area of bio/nano integration where biological components are interfaced with inorganic nanomaterials to create new devices and systems that combine the desirable properties of each system. One particular nanomaterial used in this kind of research are carbon nanotubes (CNTs). Scientists now report the integration of a CNT transistor with olfactory receptor proteins. The ultimate goal of this type of research is to transfer the sensing properties of biological molecular systems to artificial electronic devices.
Integrating biological molecules or even complex molecular machines with man-made nanoelectronic devices is one of the ultimate goals of bionanotechnology. Already there is a growing community of researchers interested in this area of bio/nano integration where biological components are interfaced with inorganic nanomaterials to create new devices and systems that combine the desirable properties of each system. One particular nanomaterial used in this kind of research are carbon nanotubes (CNTs). Scientists now report the integration of a CNT transistor with olfactory receptor proteins. The ultimate goal of this type of research is to transfer the sensing properties of biological molecular systems to artificial electronic devices.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





