Showing Spotlights 1625 - 1632 of 2779 in category All (newest first):
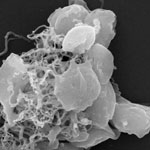 Blood platelets are the structural and chemical foundation of blood clotting and they play a vital role in minor injuries when coagulation prevents the loss of blood at the injury site. If the proper function of these platelets gets disturbed, blood clotting can lead to thrombosis, which is a leading cause of death and disability in the developed world. In view of the rapid development of nanotechnology, the impact of the newly engineered nanomaterials as an additional thrombosis risk factor is not yet known but should not be underestimated. In fact, it has been reported that carbon nanotubes induce platelet aggregation and potentiate arterial thrombosis in animal model. However, a mechanism of thrombogenic effects of carbon nanotubes was not known. Researchers have now shown that show the molecular mechanism of carbon nanotubes' induced platelets activation.
Blood platelets are the structural and chemical foundation of blood clotting and they play a vital role in minor injuries when coagulation prevents the loss of blood at the injury site. If the proper function of these platelets gets disturbed, blood clotting can lead to thrombosis, which is a leading cause of death and disability in the developed world. In view of the rapid development of nanotechnology, the impact of the newly engineered nanomaterials as an additional thrombosis risk factor is not yet known but should not be underestimated. In fact, it has been reported that carbon nanotubes induce platelet aggregation and potentiate arterial thrombosis in animal model. However, a mechanism of thrombogenic effects of carbon nanotubes was not known. Researchers have now shown that show the molecular mechanism of carbon nanotubes' induced platelets activation.
Jul 7th, 2011
 The field of single-molecule magnets is very promising, since an individual magnetic molecule represent the ultimate size limit to store and processing information. Magnetic molecules are considered very promising for spintronics since they can store a bit of information in an extremely small volume. However, in order to make magnetic molecules work, one has to find a way to measure their magnetization. Usual approaches are often very invasive and may lead to a strong perturbation of the properties of the molecules. A European research team has now designed and realized a novel hybrid spintronic nanodevice where the state of the molecule is measured 'indirectly', through a sensor coupled with the molecule.
The field of single-molecule magnets is very promising, since an individual magnetic molecule represent the ultimate size limit to store and processing information. Magnetic molecules are considered very promising for spintronics since they can store a bit of information in an extremely small volume. However, in order to make magnetic molecules work, one has to find a way to measure their magnetization. Usual approaches are often very invasive and may lead to a strong perturbation of the properties of the molecules. A European research team has now designed and realized a novel hybrid spintronic nanodevice where the state of the molecule is measured 'indirectly', through a sensor coupled with the molecule.
Jul 5th, 2011
 The efforts undertaken in developing renewable energy sources to reduce our dependence on fossil fuels include major research and investment in advanced electricity storage technologies. Among the various existing technologies, lithium batteries are considered as the most competitive power source because of their high energy density, superior power capability, design flexibility and longer lifespan. This article provides an overview of the current patent landscape of rechargeable Li-ion battery, with a focus on the recent developments on nanomaterials and nanotechnologies used for anode, cathode, and electrolyte materials, and the impact of nanomaterials on the performance of rechargeable lithium batteries. Effort has also been taken to identify key players, emerging trends and applications in this area.
The efforts undertaken in developing renewable energy sources to reduce our dependence on fossil fuels include major research and investment in advanced electricity storage technologies. Among the various existing technologies, lithium batteries are considered as the most competitive power source because of their high energy density, superior power capability, design flexibility and longer lifespan. This article provides an overview of the current patent landscape of rechargeable Li-ion battery, with a focus on the recent developments on nanomaterials and nanotechnologies used for anode, cathode, and electrolyte materials, and the impact of nanomaterials on the performance of rechargeable lithium batteries. Effort has also been taken to identify key players, emerging trends and applications in this area.
Jul 1st, 2011
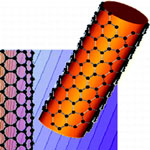 There has been tremendous interest in cloaking technology using metamaterials and there are two exciting possibilities to obtain drastic scattering reduction from moderately-sized objects. One is the concept of plasmonic cloaking, which is based on the use of a thin metamaterial cover to suppress the scattering from a passive object. The second one is the concept of mantle cloaking, which is based on a simple impedance surface to achieve similar effects. Now, new research shows that even a single layer of atoms, with the exciting conductivity properties of graphene, may achieve similar functionality in planar and cylindrical geometries. Once realized, this concept represents the 'thinnest' possible cloak, operating in the THz spectrum.
There has been tremendous interest in cloaking technology using metamaterials and there are two exciting possibilities to obtain drastic scattering reduction from moderately-sized objects. One is the concept of plasmonic cloaking, which is based on the use of a thin metamaterial cover to suppress the scattering from a passive object. The second one is the concept of mantle cloaking, which is based on a simple impedance surface to achieve similar effects. Now, new research shows that even a single layer of atoms, with the exciting conductivity properties of graphene, may achieve similar functionality in planar and cylindrical geometries. Once realized, this concept represents the 'thinnest' possible cloak, operating in the THz spectrum.
Jun 30th, 2011
 Nanoindentation is derived from the classical hardness test but is carried out on a much smaller scale. It can be used to determine the hardness of thin layers as well as material properties such as elasticity, stiffness, plasticity, and tensile strength, or fracture toughness of small objects and microsystems in fields such as biotechnology. These measurements involve applying a small force to a sample using a sharp probe and measuring the resultant penetration depth. The measured value is used to calculate the contact area and hence the particular property of the sample material. Both the method of force application and the geometry of the indentation tip can be adjusted to suit the particular application.
Nanoindentation is derived from the classical hardness test but is carried out on a much smaller scale. It can be used to determine the hardness of thin layers as well as material properties such as elasticity, stiffness, plasticity, and tensile strength, or fracture toughness of small objects and microsystems in fields such as biotechnology. These measurements involve applying a small force to a sample using a sharp probe and measuring the resultant penetration depth. The measured value is used to calculate the contact area and hence the particular property of the sample material. Both the method of force application and the geometry of the indentation tip can be adjusted to suit the particular application.
Jun 29th, 2011
 Notwithstanding all the buzz about renewable energy sources, the dirty facts are that coal accounts for 41% of electricity production worldwide. Since, realistically, coal will be a mainstay of electricity generation for many years to come, research into more environmentally friendly use of coal energy is picking up steam. One technology for more efficient power production centers around the solid oxide fuel cell (SOFC). Especially gasified carbon fuel cells offer great prospects for the most efficient utilization of a wide variety of carbonaceous solids fuels, including coal, biomass, and municipal solid waste. Researchers have now developed a self-cleaning technique that could allow solid oxide fuel cells to be powered directly by coal gas at operating temperatures as low as 750 degrees Celsius.
Notwithstanding all the buzz about renewable energy sources, the dirty facts are that coal accounts for 41% of electricity production worldwide. Since, realistically, coal will be a mainstay of electricity generation for many years to come, research into more environmentally friendly use of coal energy is picking up steam. One technology for more efficient power production centers around the solid oxide fuel cell (SOFC). Especially gasified carbon fuel cells offer great prospects for the most efficient utilization of a wide variety of carbonaceous solids fuels, including coal, biomass, and municipal solid waste. Researchers have now developed a self-cleaning technique that could allow solid oxide fuel cells to be powered directly by coal gas at operating temperatures as low as 750 degrees Celsius.
Jun 28th, 2011
 One enabling technology for printed and flexible electronics devices is the use of a special silver nanoparticle ink that allow the patterning of silver microelectrodes by omnidirectional printing. Taking an important step towards enabling desktop manufacturing - or personal fabrication - using very low cost, ubiquitous printing tools, researchers have now demonstrated a pen-on-paper approach as a low-cost, portable fabrication route for printed electronic and optoelectronic devices. The team demonstrates the fabrication of electronic art, flexible displays, conductive text, and radio frequency antennas with their technique. The printed features can withstand repeated bending and folding while maintaining high conductivity.
One enabling technology for printed and flexible electronics devices is the use of a special silver nanoparticle ink that allow the patterning of silver microelectrodes by omnidirectional printing. Taking an important step towards enabling desktop manufacturing - or personal fabrication - using very low cost, ubiquitous printing tools, researchers have now demonstrated a pen-on-paper approach as a low-cost, portable fabrication route for printed electronic and optoelectronic devices. The team demonstrates the fabrication of electronic art, flexible displays, conductive text, and radio frequency antennas with their technique. The printed features can withstand repeated bending and folding while maintaining high conductivity.
Jun 27th, 2011
 Among various technologies, reverse osmosis membranes have been widely used for water reclamation. However, external energy required and high operational pressure used make reverse osmosis membrane water reclamation processes energy intensive - not exactly an advantage given the rising cost of energy and the negative climate impact of fossil fuels. Today, forward osmosis is a well-recognized osmotic process for producing clean water with a bright future as it uses a natural phenomenon and does not require any operational pressure hence it saves large amount of energy compared with reverse osmosis process. Researchers now describe a novel forward osmosis membrane that presents remarkable properties superior over conventional membrane support layers.
Among various technologies, reverse osmosis membranes have been widely used for water reclamation. However, external energy required and high operational pressure used make reverse osmosis membrane water reclamation processes energy intensive - not exactly an advantage given the rising cost of energy and the negative climate impact of fossil fuels. Today, forward osmosis is a well-recognized osmotic process for producing clean water with a bright future as it uses a natural phenomenon and does not require any operational pressure hence it saves large amount of energy compared with reverse osmosis process. Researchers now describe a novel forward osmosis membrane that presents remarkable properties superior over conventional membrane support layers.
Jun 24th, 2011
 Blood platelets are the structural and chemical foundation of blood clotting and they play a vital role in minor injuries when coagulation prevents the loss of blood at the injury site. If the proper function of these platelets gets disturbed, blood clotting can lead to thrombosis, which is a leading cause of death and disability in the developed world. In view of the rapid development of nanotechnology, the impact of the newly engineered nanomaterials as an additional thrombosis risk factor is not yet known but should not be underestimated. In fact, it has been reported that carbon nanotubes induce platelet aggregation and potentiate arterial thrombosis in animal model. However, a mechanism of thrombogenic effects of carbon nanotubes was not known. Researchers have now shown that show the molecular mechanism of carbon nanotubes' induced platelets activation.
Blood platelets are the structural and chemical foundation of blood clotting and they play a vital role in minor injuries when coagulation prevents the loss of blood at the injury site. If the proper function of these platelets gets disturbed, blood clotting can lead to thrombosis, which is a leading cause of death and disability in the developed world. In view of the rapid development of nanotechnology, the impact of the newly engineered nanomaterials as an additional thrombosis risk factor is not yet known but should not be underestimated. In fact, it has been reported that carbon nanotubes induce platelet aggregation and potentiate arterial thrombosis in animal model. However, a mechanism of thrombogenic effects of carbon nanotubes was not known. Researchers have now shown that show the molecular mechanism of carbon nanotubes' induced platelets activation.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





