Showing Spotlights 169 - 176 of 544 in category All (newest first):
 Nanotechnology is becoming a crucial driving force behind innovation in medicine and healthcare, with a range of advances including nanoscale therapeutics, biosensors, implantable devices, drug delivery systems, and imaging technologies. This article provides a comprehensive overview of healthcare advances that may be possible through nanotechnology, ranging from fitness monitoring, prevention, diagnosis to therapy, and everything in between.
Nanotechnology is becoming a crucial driving force behind innovation in medicine and healthcare, with a range of advances including nanoscale therapeutics, biosensors, implantable devices, drug delivery systems, and imaging technologies. This article provides a comprehensive overview of healthcare advances that may be possible through nanotechnology, ranging from fitness monitoring, prevention, diagnosis to therapy, and everything in between.
Jun 13th, 2017
 Inspired by octopus arms, researchers demonstrate an uncomplicated and scalable templating technology for fabricating nanosuckers on PDMS substrates, using a scalable spin-coating technology. As the nanosuckers are pressed against a substrate, the flexible nanosuckers confirm to the substrate and deform by releasing the internal air between nanosuckers and the substrate, forming a seal and generating an adhesion force. The nanosucker adhesion is maintained over multiple contact cycles on both wet and dry surfaces.
Inspired by octopus arms, researchers demonstrate an uncomplicated and scalable templating technology for fabricating nanosuckers on PDMS substrates, using a scalable spin-coating technology. As the nanosuckers are pressed against a substrate, the flexible nanosuckers confirm to the substrate and deform by releasing the internal air between nanosuckers and the substrate, forming a seal and generating an adhesion force. The nanosucker adhesion is maintained over multiple contact cycles on both wet and dry surfaces.
May 25th, 2017
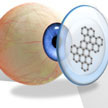 Several research projects are working on reinventing the contact lens as a smart electronic device that, for instance, works as a self-powered biosensor for various point-of-care monitoring and wireless biomedical sensing. n addition to sensors, researchers are devising numerous applications for smart contact lenses, ranging from drug delivery systems to protection from electromagnetic wave damage. An application closer to contact lenses' original function, graphene can change the focal length of a polymeric soft contact lens in order to adjust near- and farsightedness.
Several research projects are working on reinventing the contact lens as a smart electronic device that, for instance, works as a self-powered biosensor for various point-of-care monitoring and wireless biomedical sensing. n addition to sensors, researchers are devising numerous applications for smart contact lenses, ranging from drug delivery systems to protection from electromagnetic wave damage. An application closer to contact lenses' original function, graphene can change the focal length of a polymeric soft contact lens in order to adjust near- and farsightedness.
May 22nd, 2017
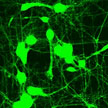 Growing neurons outside the brain but with predictable synaptic connectivity between other neurons could provide for an efficient platform for fundamental research and design of neuroprosthetics. Various neuromorphic engineering research efforts are underway to do this. A team now has built a Brain-on-a-chip where they demonstrated guided growth of neurons on semiconductor nanowire scaffolds. Providing an environment were scientists could study a less complex neuronal circuit as opposed to a fully functioning circuit in a living mammalian brain will open up a new experimental paradigm of understanding how the neurons are influenced by the mechanical properties of the brain as they grow and form circuits.
Growing neurons outside the brain but with predictable synaptic connectivity between other neurons could provide for an efficient platform for fundamental research and design of neuroprosthetics. Various neuromorphic engineering research efforts are underway to do this. A team now has built a Brain-on-a-chip where they demonstrated guided growth of neurons on semiconductor nanowire scaffolds. Providing an environment were scientists could study a less complex neuronal circuit as opposed to a fully functioning circuit in a living mammalian brain will open up a new experimental paradigm of understanding how the neurons are influenced by the mechanical properties of the brain as they grow and form circuits.
May 12th, 2017
 Immunotherapy has become an important part of treating some types of cancer. It uses certain parts of a person's immune system to fight the cancer. Usually this is done by administering immune system components, such as man-made immune system proteins. In recent years, nanotechnology has played an increasingly important role in pursuing efficient vaccine delivery in cancer immunotherapy. This article discusses vaccine delivery by synthetic nanoparticles or naturally derived nanoparticles for cancer immunotherapy.
Immunotherapy has become an important part of treating some types of cancer. It uses certain parts of a person's immune system to fight the cancer. Usually this is done by administering immune system components, such as man-made immune system proteins. In recent years, nanotechnology has played an increasingly important role in pursuing efficient vaccine delivery in cancer immunotherapy. This article discusses vaccine delivery by synthetic nanoparticles or naturally derived nanoparticles for cancer immunotherapy.
May 4th, 2017
 Researchers show how spermatozoa can be useful parts of microdevices: As biocompatible propulsion source, but also entailing other functionalities such as their natural destiny for fertilization, their ability to respond to stimuli, or their ability to take up drugs open up fascinating new applications. They demonstrate first examples of using sperm cells as robotic components. The so-called spermbots are also systems that enable biophysical studies, e.g. of sperm motion in confinement.
Researchers show how spermatozoa can be useful parts of microdevices: As biocompatible propulsion source, but also entailing other functionalities such as their natural destiny for fertilization, their ability to respond to stimuli, or their ability to take up drugs open up fascinating new applications. They demonstrate first examples of using sperm cells as robotic components. The so-called spermbots are also systems that enable biophysical studies, e.g. of sperm motion in confinement.
May 3rd, 2017
 Molecular magnets or single molecule-based magnets are usually anti-ferromagnetic (non-magnetic) at room temperature, which so far has limited their use to laboratory environments. As the first successful molecular magnet in a real-world application, an interdisciplinary research group has reported a new 'exotic' molecular magnet compound - iron salen nanoparticles - which shows intrinsic magnetic nature at room temperature as well as anticancer properties.
Molecular magnets or single molecule-based magnets are usually anti-ferromagnetic (non-magnetic) at room temperature, which so far has limited their use to laboratory environments. As the first successful molecular magnet in a real-world application, an interdisciplinary research group has reported a new 'exotic' molecular magnet compound - iron salen nanoparticles - which shows intrinsic magnetic nature at room temperature as well as anticancer properties.
Apr 13th, 2017
 Recently, great progress has been made in the development of bio-hybrid devices with enhanced biological, mechanical and electrical designs. Several muscular tissue based actuators have been described and devices with cultured heart cells have also been reported to produce electrical outputs.
Now, researchers have demonstrated a novel bio-hybrid system, the 'Cell Generator'. The researchers integrated piezoelectric material with 3D-engineered living constructs for energy harvesting and electricity generation.
Recently, great progress has been made in the development of bio-hybrid devices with enhanced biological, mechanical and electrical designs. Several muscular tissue based actuators have been described and devices with cultured heart cells have also been reported to produce electrical outputs.
Now, researchers have demonstrated a novel bio-hybrid system, the 'Cell Generator'. The researchers integrated piezoelectric material with 3D-engineered living constructs for energy harvesting and electricity generation.
Mar 31st, 2017
 Nanotechnology is becoming a crucial driving force behind innovation in medicine and healthcare, with a range of advances including nanoscale therapeutics, biosensors, implantable devices, drug delivery systems, and imaging technologies. This article provides a comprehensive overview of healthcare advances that may be possible through nanotechnology, ranging from fitness monitoring, prevention, diagnosis to therapy, and everything in between.
Nanotechnology is becoming a crucial driving force behind innovation in medicine and healthcare, with a range of advances including nanoscale therapeutics, biosensors, implantable devices, drug delivery systems, and imaging technologies. This article provides a comprehensive overview of healthcare advances that may be possible through nanotechnology, ranging from fitness monitoring, prevention, diagnosis to therapy, and everything in between.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





