Showing Spotlights 281 - 288 of 544 in category All (newest first):
 The heating properties of iron oxide nanoparticles have been exploited through the years for use in cancer therapy, gene regulation, and temperature responsive valves. These applications have demonstrated the versatility of iron oxide nanoparticles, but they had rarely, if ever, been used to enhance the activity of thermophilic enzymes. Thermophilic enzymes are highly stable biomolecular systems that are excellent tools due to their thermostability and long-term activity for extended lifetime uses in the field and other applications. New work by researchers in the U.S. addresses the problem of remotely activating biological materials with a higher efficiency than conventional methods such as water baths or convection ovens.
The heating properties of iron oxide nanoparticles have been exploited through the years for use in cancer therapy, gene regulation, and temperature responsive valves. These applications have demonstrated the versatility of iron oxide nanoparticles, but they had rarely, if ever, been used to enhance the activity of thermophilic enzymes. Thermophilic enzymes are highly stable biomolecular systems that are excellent tools due to their thermostability and long-term activity for extended lifetime uses in the field and other applications. New work by researchers in the U.S. addresses the problem of remotely activating biological materials with a higher efficiency than conventional methods such as water baths or convection ovens.
Oct 15th, 2012
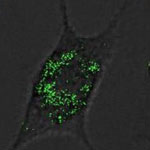 Silver nanoparticles are among the most commercialized nanomaterials due to their use as antibacterial agent in consumer products and surface coatings. Several reports have shown that silver ions from silver compounds or those that develop from nanosilver particles through contact with water are highly toxic to microorganisms such as bacteria, fungi and algae. Contributing to an incomplete and confusing picture, in the literature, silver nanoparticles are claimed as nontoxic or toxic depending on their size, concentration and surface functionalization. Researchers have now reported on possible adverse effects of the silver nanoparticles upon their release into the environment and provided novel insights into the possible applications of silver nanoparticles in nanomedicine by discussing their p53 gene related cell death profiles.
Silver nanoparticles are among the most commercialized nanomaterials due to their use as antibacterial agent in consumer products and surface coatings. Several reports have shown that silver ions from silver compounds or those that develop from nanosilver particles through contact with water are highly toxic to microorganisms such as bacteria, fungi and algae. Contributing to an incomplete and confusing picture, in the literature, silver nanoparticles are claimed as nontoxic or toxic depending on their size, concentration and surface functionalization. Researchers have now reported on possible adverse effects of the silver nanoparticles upon their release into the environment and provided novel insights into the possible applications of silver nanoparticles in nanomedicine by discussing their p53 gene related cell death profiles.
Sep 12th, 2012
 Nanotechnology is gaining significant interest in plant sciences with research focusing on investigating plant genomics and gene function as well as improving crop species. The impact on agriculture could be dramatic. Most of the work done with nanoparticles in plant sciences relies on a passive uptake of nanoparticles by plant cells - a process that cannot be controlled. Plant cells differ from animal cells in several aspects, a major one being that they possess a wall surrounding them to provide, among other things, mechanical and structural support. Researchers are commonly using surface-functionalized silica nanoparticles as nonviral nanocarriers for experimental drug and DNA delivery into animal cells but their use with plants so far was limited due to the barriers posed by the cell wall. Following up on previous work, researchers have made this process more efficient and introduced a much more challenging biomolecule to plant cells - proteins.
Nanotechnology is gaining significant interest in plant sciences with research focusing on investigating plant genomics and gene function as well as improving crop species. The impact on agriculture could be dramatic. Most of the work done with nanoparticles in plant sciences relies on a passive uptake of nanoparticles by plant cells - a process that cannot be controlled. Plant cells differ from animal cells in several aspects, a major one being that they possess a wall surrounding them to provide, among other things, mechanical and structural support. Researchers are commonly using surface-functionalized silica nanoparticles as nonviral nanocarriers for experimental drug and DNA delivery into animal cells but their use with plants so far was limited due to the barriers posed by the cell wall. Following up on previous work, researchers have made this process more efficient and introduced a much more challenging biomolecule to plant cells - proteins.
Sep 10th, 2012
 Thanks to nanotechnologies, in particular nanoelectronics, the medical sector is about to undergo deep changes by exploiting the traditional strengths of the semiconductor industry - miniaturization and integration. While conventional electronics have already found many applications in biomedicine - medical monitoring of vital signals, biophysical studies of excitable tissues, implantable electrodes for brain stimulation, pacemakers, and limb stimulation - the use of nanomaterials and nanoscale applications will bring a further push towards implanted electronics in the human body. A new perspective article provides an overview of nanoelectronics' potential in the biomedical sciences.
Thanks to nanotechnologies, in particular nanoelectronics, the medical sector is about to undergo deep changes by exploiting the traditional strengths of the semiconductor industry - miniaturization and integration. While conventional electronics have already found many applications in biomedicine - medical monitoring of vital signals, biophysical studies of excitable tissues, implantable electrodes for brain stimulation, pacemakers, and limb stimulation - the use of nanomaterials and nanoscale applications will bring a further push towards implanted electronics in the human body. A new perspective article provides an overview of nanoelectronics' potential in the biomedical sciences.
Aug 16th, 2012
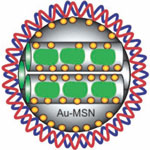
 Nitrogen-doped carbon nanotubes (CNTs) have been extensively investigated for fuel cell applications due to their excellent electrocatalytic properties. However, their biomedical applications were comparatively less investigated despite reports of their better biocompatibility. When considering carbon nanotubes for drug delivery applications, it is desirable to develop strategies that allow utilize their hollow inner cavities for maximum loading capacity. Small size and facile surface modification are also preferable with regard to their biomedical compatibility. Nitrogen-doped CNTs have been already previously demonstrated to have better biocompatibility and mitigated cytotoxicity as compared to traditional undoped pristine CNTs. Taking advantage of this, researchers used nitrogen doping of CNTs which resulted in formation of cup-shaped compartments in CNTs uniquely suitable for encapsulation. The resulting nitrogen-doped carbon nanotube cups can be corked by gold nanoparticles to form enclosed nanocapsules.
Nitrogen-doped carbon nanotubes (CNTs) have been extensively investigated for fuel cell applications due to their excellent electrocatalytic properties. However, their biomedical applications were comparatively less investigated despite reports of their better biocompatibility. When considering carbon nanotubes for drug delivery applications, it is desirable to develop strategies that allow utilize their hollow inner cavities for maximum loading capacity. Small size and facile surface modification are also preferable with regard to their biomedical compatibility. Nitrogen-doped CNTs have been already previously demonstrated to have better biocompatibility and mitigated cytotoxicity as compared to traditional undoped pristine CNTs. Taking advantage of this, researchers used nitrogen doping of CNTs which resulted in formation of cup-shaped compartments in CNTs uniquely suitable for encapsulation. The resulting nitrogen-doped carbon nanotube cups can be corked by gold nanoparticles to form enclosed nanocapsules.
Aug 2nd, 2012
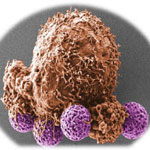 Early and accurate detection of cancer is critical for successful cancer therapies. In most cases, a tissue biopsy is the initial means of making a diagnosis. With increasing accuracy, 'liquid biopsies' - where circulating tumor cells (CTCs) are isolated from blood samples - are becoming a viable complement or even alternative to invasive biopsies of metastatic tumors. CTC is of great interest for evaluating cancer dissemination, predicting patient prognosis, and also for the evaluation of therapeutic treatments. In new work, researchers describe a rapid and simple electrochemical biosensing strategy to quantify circulating tumour cells based on the simultaneous use of antibody-coated magnetic beads, which selectively bind to the cancer cells for subsequent magnetic isolation, and antibody-coated gold nanoparticles, to also selectively bind to the cancer cells for final electrochemical detection.
Early and accurate detection of cancer is critical for successful cancer therapies. In most cases, a tissue biopsy is the initial means of making a diagnosis. With increasing accuracy, 'liquid biopsies' - where circulating tumor cells (CTCs) are isolated from blood samples - are becoming a viable complement or even alternative to invasive biopsies of metastatic tumors. CTC is of great interest for evaluating cancer dissemination, predicting patient prognosis, and also for the evaluation of therapeutic treatments. In new work, researchers describe a rapid and simple electrochemical biosensing strategy to quantify circulating tumour cells based on the simultaneous use of antibody-coated magnetic beads, which selectively bind to the cancer cells for subsequent magnetic isolation, and antibody-coated gold nanoparticles, to also selectively bind to the cancer cells for final electrochemical detection.
Aug 1st, 2012
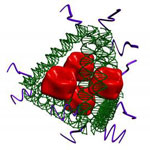 Vaccination is one of the most effective ways to prevent microbial infection. Synthetic vaccines can combine a portion of a microbe, known as an 'antigen' together with an adjuvant that stimulates the immune system. Delivering both the adjuvant and antigen to the appropriate immune cells is challenging. DNA nanotechnology may provide a solution by acting as a scaffold to co-deliver both antigen and adjuvant. However, the potential of DNA nanostructure-based vaccines has only been demonstrated in vitro. Now, a team of researchers based out of Arizona State University demonstrated that DNA nanostructures with appended adjuvants could elicit antibody production against a model antigen in mice.
Vaccination is one of the most effective ways to prevent microbial infection. Synthetic vaccines can combine a portion of a microbe, known as an 'antigen' together with an adjuvant that stimulates the immune system. Delivering both the adjuvant and antigen to the appropriate immune cells is challenging. DNA nanotechnology may provide a solution by acting as a scaffold to co-deliver both antigen and adjuvant. However, the potential of DNA nanostructure-based vaccines has only been demonstrated in vitro. Now, a team of researchers based out of Arizona State University demonstrated that DNA nanostructures with appended adjuvants could elicit antibody production against a model antigen in mice.
Jul 30th, 2012
 A major obstacle in today's chemical cancer therapies is to achieve specific drug accumulation at tumor sites and even tumor cells, because most chemotherapeutic agents are insoluble and instable, and can not differentiate between diseased and healthy cells. This often leads to severe adverse effects on healthy tissues and limits the maximum dose that can safely administered to patients. This issue becomes more serious in the case of some highly effective therapeutic agents, given that most potent anticancer drugs exhibit acute toxicity and narrow therapeutic window, and the clinical utility may not be possible without a powerful delivery systems equipped with smart properties that can allow them to precisely access the target tissues and cells. Targeted delivery approaches using nanomedicines will be able to provide solutions to these problems.
A major obstacle in today's chemical cancer therapies is to achieve specific drug accumulation at tumor sites and even tumor cells, because most chemotherapeutic agents are insoluble and instable, and can not differentiate between diseased and healthy cells. This often leads to severe adverse effects on healthy tissues and limits the maximum dose that can safely administered to patients. This issue becomes more serious in the case of some highly effective therapeutic agents, given that most potent anticancer drugs exhibit acute toxicity and narrow therapeutic window, and the clinical utility may not be possible without a powerful delivery systems equipped with smart properties that can allow them to precisely access the target tissues and cells. Targeted delivery approaches using nanomedicines will be able to provide solutions to these problems.
Jul 25th, 2012
 The heating properties of iron oxide nanoparticles have been exploited through the years for use in cancer therapy, gene regulation, and temperature responsive valves. These applications have demonstrated the versatility of iron oxide nanoparticles, but they had rarely, if ever, been used to enhance the activity of thermophilic enzymes. Thermophilic enzymes are highly stable biomolecular systems that are excellent tools due to their thermostability and long-term activity for extended lifetime uses in the field and other applications. New work by researchers in the U.S. addresses the problem of remotely activating biological materials with a higher efficiency than conventional methods such as water baths or convection ovens.
The heating properties of iron oxide nanoparticles have been exploited through the years for use in cancer therapy, gene regulation, and temperature responsive valves. These applications have demonstrated the versatility of iron oxide nanoparticles, but they had rarely, if ever, been used to enhance the activity of thermophilic enzymes. Thermophilic enzymes are highly stable biomolecular systems that are excellent tools due to their thermostability and long-term activity for extended lifetime uses in the field and other applications. New work by researchers in the U.S. addresses the problem of remotely activating biological materials with a higher efficiency than conventional methods such as water baths or convection ovens.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





