Showing Spotlights 289 - 296 of 544 in category All (newest first):
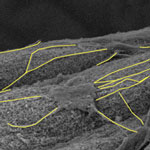 Carbon nanotubes, like the nervous cells of our brain, are excellent electrical signal conductors and can form intimate mechanical contacts with cellular membranes, thereby establishing a functional link to neuronal structures. There is a growing body of research on using nanomaterials in neural engineering. This emerging field of 'nanoneuroscience' explores and utilizes the potential of carbon nanotubes (CNTs) for fabricating durable, robust substrates, scaffolds and interfacing devices that change how researchers approach neuroscience. One area of neuron-CNT interaction that is now being probed is the effect of CNTs on neural stem cells. In new work by a team of scientists from Taiwan, it was found that electrical stimulation significantly promotes the early differentiation and maturation of neural stem cells.
Carbon nanotubes, like the nervous cells of our brain, are excellent electrical signal conductors and can form intimate mechanical contacts with cellular membranes, thereby establishing a functional link to neuronal structures. There is a growing body of research on using nanomaterials in neural engineering. This emerging field of 'nanoneuroscience' explores and utilizes the potential of carbon nanotubes (CNTs) for fabricating durable, robust substrates, scaffolds and interfacing devices that change how researchers approach neuroscience. One area of neuron-CNT interaction that is now being probed is the effect of CNTs on neural stem cells. In new work by a team of scientists from Taiwan, it was found that electrical stimulation significantly promotes the early differentiation and maturation of neural stem cells.
Jul 23rd, 2012
 Breath analysis of exhaled breath condensate has been increasingly recognized as a promising diagnostic method to link specific gaseous components in human breath to medical conditions and exposure to chemical compounds. Sampling breath is also much less invasive than testing blood, can be done very quickly, and creates as good as no biohazard waste. Studies have shown that exhaled breath from a flu patient contains influenza viruses but, although the use of silicon nanowire (SiNW) sensors for virus detection is not new, so far no studies have been conducted to apply silicon nanowire technology to the diagnosis of flu. Now, new research suggests that a SiNW sensor device, when calibrated by virus standards and exhaled breath condensate controls, can be reliably applied to the diagnosis of flu in a clinical setting with two orders of magnitude less time compared to the gold standard method RT-qPCR.
Breath analysis of exhaled breath condensate has been increasingly recognized as a promising diagnostic method to link specific gaseous components in human breath to medical conditions and exposure to chemical compounds. Sampling breath is also much less invasive than testing blood, can be done very quickly, and creates as good as no biohazard waste. Studies have shown that exhaled breath from a flu patient contains influenza viruses but, although the use of silicon nanowire (SiNW) sensors for virus detection is not new, so far no studies have been conducted to apply silicon nanowire technology to the diagnosis of flu. Now, new research suggests that a SiNW sensor device, when calibrated by virus standards and exhaled breath condensate controls, can be reliably applied to the diagnosis of flu in a clinical setting with two orders of magnitude less time compared to the gold standard method RT-qPCR.
Jun 29th, 2012
 Many nanotechnology projects require some form of nanopatterning technique for fabricating the devices, structures and surfaces required in fields ranging from electronics to photonics, security, biotechnology and medicine. Although they may not be visible to the naked eye, the nanometer-sized trenches, ridges, curves and grooves of these patterns and surfaces have a very visible impact. Researchers have developed a wide range of nanopatterning techniques, from top-down methods such as nanoimprint, e-beam or UV lithography to bottom-up techniques such as transfer nanolithography or nanopositioning on DNA or protein scaffolds. A novel technique uses a biofunctionalization approach based on resist-less electron-beam-induced deposition of carbon-containing nanofeatures, that has been developed into a universal biofunctionalization platform. This unique ability can be exploited for biological experiments, where cells respond to the nanoscale density of activating molecules such as antibodies.
Many nanotechnology projects require some form of nanopatterning technique for fabricating the devices, structures and surfaces required in fields ranging from electronics to photonics, security, biotechnology and medicine. Although they may not be visible to the naked eye, the nanometer-sized trenches, ridges, curves and grooves of these patterns and surfaces have a very visible impact. Researchers have developed a wide range of nanopatterning techniques, from top-down methods such as nanoimprint, e-beam or UV lithography to bottom-up techniques such as transfer nanolithography or nanopositioning on DNA or protein scaffolds. A novel technique uses a biofunctionalization approach based on resist-less electron-beam-induced deposition of carbon-containing nanofeatures, that has been developed into a universal biofunctionalization platform. This unique ability can be exploited for biological experiments, where cells respond to the nanoscale density of activating molecules such as antibodies.
Jun 26th, 2012
 Researchers in Israel have found a unique way to affect cancer cells in a rather controllable manner. For example, they can kill these cells in various ways, or they can make them fuse together, as they like. They do so by using functionalized, 20 nm in diameter gold nanospheres and specific, intense laser pulses with a visible wavelength tuned to the plasmonic resonance of those particles. This technique may have an impact on various technologies which require sophisticated cell manipulation for therapeutic and drug development applications. What is unique about this technique is that it is highly controllable and repeatable. The structural stability of the nanospheres even after the strongest laser illumination allows them to deliver the effect over and over again, until the desired result is obtained.
Researchers in Israel have found a unique way to affect cancer cells in a rather controllable manner. For example, they can kill these cells in various ways, or they can make them fuse together, as they like. They do so by using functionalized, 20 nm in diameter gold nanospheres and specific, intense laser pulses with a visible wavelength tuned to the plasmonic resonance of those particles. This technique may have an impact on various technologies which require sophisticated cell manipulation for therapeutic and drug development applications. What is unique about this technique is that it is highly controllable and repeatable. The structural stability of the nanospheres even after the strongest laser illumination allows them to deliver the effect over and over again, until the desired result is obtained.
Jun 15th, 2012
 Burn injuries are one of the major global health problems. Every year 195,000 people from all over the world die because of fire alone. A burn injury may damage some or all skin layers and is caused by a hot solid, a hot liquid, or a flame. However, injuries related to electricity, radioactivity, ultraviolet radiation, chemicals and respiratory damage due to smoking are also considered as burn injuries. Besides cleaning the wound and applying various topical anti-microbial agents, wound dressings could be an effective solution in preventing microbial infections for burn care. The suitability of a burn wound dressing depends on a burn type. Conventional dressings are not efficient enough to induce haemostasis, adherence and in holding a moist environment around wound. Due to the advances in the field of nanotechnology, it is now possible to design nanofiber-based wound dressings where an electrospun-nanofibrous layer is applied to a basic support fabric material.
Burn injuries are one of the major global health problems. Every year 195,000 people from all over the world die because of fire alone. A burn injury may damage some or all skin layers and is caused by a hot solid, a hot liquid, or a flame. However, injuries related to electricity, radioactivity, ultraviolet radiation, chemicals and respiratory damage due to smoking are also considered as burn injuries. Besides cleaning the wound and applying various topical anti-microbial agents, wound dressings could be an effective solution in preventing microbial infections for burn care. The suitability of a burn wound dressing depends on a burn type. Conventional dressings are not efficient enough to induce haemostasis, adherence and in holding a moist environment around wound. Due to the advances in the field of nanotechnology, it is now possible to design nanofiber-based wound dressings where an electrospun-nanofibrous layer is applied to a basic support fabric material.
Jun 13th, 2012
 One type of biomolecules, enzymes, regulate almost all chemical reactions involved in numerous biological processes in living organisms and are also widely used in research and industry. Regulation of enzyme activity and stability is very important and has always attracted great attention. Various enzyme regulators, ranging from proteins, peptides, and synthetic organic molecules, have been discovered. Recently, nanomaterials evolve as promising alternatives for enzyme modulation. Nanomaterials provide large surface areas for biomolecule adsorption and can be engineered to present multiple surface functional groups for interacting with biomolecules, such as enzymes and/or their substrates. In a recent study, scientists started to explore the interactions between functionalized graphene oxide and serine proteases, a large family of enzymes with important biomedical and industrial applications.
One type of biomolecules, enzymes, regulate almost all chemical reactions involved in numerous biological processes in living organisms and are also widely used in research and industry. Regulation of enzyme activity and stability is very important and has always attracted great attention. Various enzyme regulators, ranging from proteins, peptides, and synthetic organic molecules, have been discovered. Recently, nanomaterials evolve as promising alternatives for enzyme modulation. Nanomaterials provide large surface areas for biomolecule adsorption and can be engineered to present multiple surface functional groups for interacting with biomolecules, such as enzymes and/or their substrates. In a recent study, scientists started to explore the interactions between functionalized graphene oxide and serine proteases, a large family of enzymes with important biomedical and industrial applications.
Jun 7th, 2012
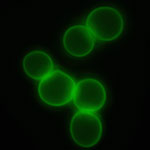 Modifying living cell by coating them with a nanolayer of functional materials in order to provide them with new structural and functional features has developed into a popular research area for bionanotechnology researchers. In contrast to genetic manipulation techniques, here the functionality of a cell is modified simply by attaching polymers or nanoparticles to the cell's surface. A recent Perspective paper covers the most interesting and promising work in this area and presents an outlook the major potential future directions. The article focuses on on cell encapsulation with Layer-by-Layer (LbL) self-assembly via sequential adsorption of oppositely charged components: polyelectrolytes, nanoparticles, and proteins.
Modifying living cell by coating them with a nanolayer of functional materials in order to provide them with new structural and functional features has developed into a popular research area for bionanotechnology researchers. In contrast to genetic manipulation techniques, here the functionality of a cell is modified simply by attaching polymers or nanoparticles to the cell's surface. A recent Perspective paper covers the most interesting and promising work in this area and presents an outlook the major potential future directions. The article focuses on on cell encapsulation with Layer-by-Layer (LbL) self-assembly via sequential adsorption of oppositely charged components: polyelectrolytes, nanoparticles, and proteins.
May 29th, 2012
 Researchers in China showed that nanoparticles smaller than 10 nm in diameter accumulate more efficiently and penetrate more deeply in tumors relative to their larger counterparts. Their findings have significant implications for the development of nanomaterials to diagnose and treat cancer. The enhanced tumor accumulation of the ultrasmall nanoparticles may be due, at least in part, to their prolonged blood circulation time. In contrast, most nanomaterials that enter the blood are rapidly cleared by tissue-resident macrophages in the liver and spleen. Ultimately, the ability of ultrasmall nanoparticles to diffuse deep within the tumor bulk may enable the design of nanoparticles that can carry therapeutic and diagnostic agents more efficiently into tumors.
Researchers in China showed that nanoparticles smaller than 10 nm in diameter accumulate more efficiently and penetrate more deeply in tumors relative to their larger counterparts. Their findings have significant implications for the development of nanomaterials to diagnose and treat cancer. The enhanced tumor accumulation of the ultrasmall nanoparticles may be due, at least in part, to their prolonged blood circulation time. In contrast, most nanomaterials that enter the blood are rapidly cleared by tissue-resident macrophages in the liver and spleen. Ultimately, the ability of ultrasmall nanoparticles to diffuse deep within the tumor bulk may enable the design of nanoparticles that can carry therapeutic and diagnostic agents more efficiently into tumors.
May 28th, 2012
 Carbon nanotubes, like the nervous cells of our brain, are excellent electrical signal conductors and can form intimate mechanical contacts with cellular membranes, thereby establishing a functional link to neuronal structures. There is a growing body of research on using nanomaterials in neural engineering. This emerging field of 'nanoneuroscience' explores and utilizes the potential of carbon nanotubes (CNTs) for fabricating durable, robust substrates, scaffolds and interfacing devices that change how researchers approach neuroscience. One area of neuron-CNT interaction that is now being probed is the effect of CNTs on neural stem cells. In new work by a team of scientists from Taiwan, it was found that electrical stimulation significantly promotes the early differentiation and maturation of neural stem cells.
Carbon nanotubes, like the nervous cells of our brain, are excellent electrical signal conductors and can form intimate mechanical contacts with cellular membranes, thereby establishing a functional link to neuronal structures. There is a growing body of research on using nanomaterials in neural engineering. This emerging field of 'nanoneuroscience' explores and utilizes the potential of carbon nanotubes (CNTs) for fabricating durable, robust substrates, scaffolds and interfacing devices that change how researchers approach neuroscience. One area of neuron-CNT interaction that is now being probed is the effect of CNTs on neural stem cells. In new work by a team of scientists from Taiwan, it was found that electrical stimulation significantly promotes the early differentiation and maturation of neural stem cells.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





