Showing Spotlights 329 - 336 of 544 in category All (newest first):
 Brain-computer interfaces, neural probes, brain implants - they all require intensive in vivo studies on how to best combine inorganic electronics with organic neurons. Currently, most neural culture studies suffer from the fact that their cells migrate on a flat surface and are directly exposed to the culture solution, which do not reflect the microenvironment in vivo - neurons, though, can alter their behaviors dramatically in response to the environment change. Ideal cultures, therefore, should mimic the native neural microenvironment to capture the normal cell behavior. This has motivated a group of researchers to come up with this a semiconductor nanomembrane tube approach, which provides a 3D confinement that can potentially isolate the cells from the culture solution. Since these tubes are made from a semiconductor material, it means that they can be integrated with electronic functionalities such as voltage sensors.
Brain-computer interfaces, neural probes, brain implants - they all require intensive in vivo studies on how to best combine inorganic electronics with organic neurons. Currently, most neural culture studies suffer from the fact that their cells migrate on a flat surface and are directly exposed to the culture solution, which do not reflect the microenvironment in vivo - neurons, though, can alter their behaviors dramatically in response to the environment change. Ideal cultures, therefore, should mimic the native neural microenvironment to capture the normal cell behavior. This has motivated a group of researchers to come up with this a semiconductor nanomembrane tube approach, which provides a 3D confinement that can potentially isolate the cells from the culture solution. Since these tubes are made from a semiconductor material, it means that they can be integrated with electronic functionalities such as voltage sensors.
Mar 31st, 2011
 Regenerative medicine, in particular the area of tissue regeneration, is seeing a rapidly growing field of novel biomaterials that can act as bioactive scaffolds that induce tissue regeneration; that is in contrast to the more traditional concept of passively accepted implant materials. In order to present biological stimuli to the physiological environment and trigger tissue repair, optimal integration of synthetic biomaterials within the surrounding tissue is of paramount importance. In that respect, hydrogels made from biodegradable polymers are ideal candidates since they are generally biocompatible, biodegradable, and, in some cases, injectable. New research has provided firm evidence for a feasible bottom-up approach for the preparation of injectable gels by employing oppositely charged gelatin nanospheres as building blocks.
Regenerative medicine, in particular the area of tissue regeneration, is seeing a rapidly growing field of novel biomaterials that can act as bioactive scaffolds that induce tissue regeneration; that is in contrast to the more traditional concept of passively accepted implant materials. In order to present biological stimuli to the physiological environment and trigger tissue repair, optimal integration of synthetic biomaterials within the surrounding tissue is of paramount importance. In that respect, hydrogels made from biodegradable polymers are ideal candidates since they are generally biocompatible, biodegradable, and, in some cases, injectable. New research has provided firm evidence for a feasible bottom-up approach for the preparation of injectable gels by employing oppositely charged gelatin nanospheres as building blocks.
Mar 30th, 2011
 In the decades-old quest to build artificial muscles, many materials have been investigated with regard to their suitability for actuator application (the ability of a material to reversibly change dimensions under the influence of various stimuli). Recently, hope in obtaining improved artificial muscles was revitalized by the discovery of the extraordinary mechanical and electrical properties of graphene and its chemical derivatives. A new study shows that electrical heating of graphene oxide nanoribbons mats results in reversible generation of 1.6% stress and maximum work capacity of about 40 J/kg, which is similar to that of natural muscle. Unlike previous studies, the team obtained the graphene material by chemical unzipping of multi-walled carbon nanotubes.
In the decades-old quest to build artificial muscles, many materials have been investigated with regard to their suitability for actuator application (the ability of a material to reversibly change dimensions under the influence of various stimuli). Recently, hope in obtaining improved artificial muscles was revitalized by the discovery of the extraordinary mechanical and electrical properties of graphene and its chemical derivatives. A new study shows that electrical heating of graphene oxide nanoribbons mats results in reversible generation of 1.6% stress and maximum work capacity of about 40 J/kg, which is similar to that of natural muscle. Unlike previous studies, the team obtained the graphene material by chemical unzipping of multi-walled carbon nanotubes.
Mar 24th, 2011
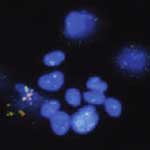 Multifunctional nanomaterials have become widely researched in nanomedicine with the goal of developing highly accurate probes for detecting and isolating cancer cells. Of particular interest here are magnetic nanoparticles, which offer the capability of cell isolation from original or enriched samples without the use of centrifugation or filtration. In particular, the combination of fluorescent quantum dots and magnetic nanoparticles into single nanospheres to obtain fluorescent-magnetic bifunctional nanospheres has created the potential for broader applications in biomedicine and in clinical diagnosis. In new work, researchers have now expanded this technique to multiplexed assays. They demonstrated the ability to detect and collect multiple types of cancer cells, such as leukemia cells and prostate cancer cells, from mixed samples within 25 minutes by using a magnet and an ordinary fluorescence microscope.
Multifunctional nanomaterials have become widely researched in nanomedicine with the goal of developing highly accurate probes for detecting and isolating cancer cells. Of particular interest here are magnetic nanoparticles, which offer the capability of cell isolation from original or enriched samples without the use of centrifugation or filtration. In particular, the combination of fluorescent quantum dots and magnetic nanoparticles into single nanospheres to obtain fluorescent-magnetic bifunctional nanospheres has created the potential for broader applications in biomedicine and in clinical diagnosis. In new work, researchers have now expanded this technique to multiplexed assays. They demonstrated the ability to detect and collect multiple types of cancer cells, such as leukemia cells and prostate cancer cells, from mixed samples within 25 minutes by using a magnet and an ordinary fluorescence microscope.
Mar 4th, 2011
 Research to develop sensors that can rapidly detect biomarkers (associated with certain diseases such as cancer) in whole blood, ideally at the point of care, and when the protein biomarker level in blood is very low (i.e. the disease is in an early stage) is being advanced by nanosensor technology. In a recent development, scientists in Spain have developed a rapid nanochannel-based immunoassay capable of the filtering and subsequent detection of proteins in whole blood without any sample preparation. This is the first time that a simple assay to detect proteins in whole blood using nanochannels has been achieved. This is a simple device and set-up that allows nanochannels to achieve such the double functionality of filtering and detection on the same platform.
Research to develop sensors that can rapidly detect biomarkers (associated with certain diseases such as cancer) in whole blood, ideally at the point of care, and when the protein biomarker level in blood is very low (i.e. the disease is in an early stage) is being advanced by nanosensor technology. In a recent development, scientists in Spain have developed a rapid nanochannel-based immunoassay capable of the filtering and subsequent detection of proteins in whole blood without any sample preparation. This is the first time that a simple assay to detect proteins in whole blood using nanochannels has been achieved. This is a simple device and set-up that allows nanochannels to achieve such the double functionality of filtering and detection on the same platform.
Feb 23rd, 2011
 Controlled-release drug delivery techniques use various responsive materials and stimuli such as an implanted heat source or electronic chip to trigger the drug release from the implanted reservoir. Among many actively controlled stimuli for pulsatile drug delivery, the electrical signal would be the best source because it is portable and does not need large or special equipment to trigger it. The signal can also be easily and on-demand controllable, and long cycles are possible. Furthermore, when a sensor or microchip system is combined, the feedback and remote control outside the body is possible. Researchers have now developed a new drug delivery device for pulsatile and on-demand drug release based on electrically actuatable nanoporous membranes made of polypyrrole.
Controlled-release drug delivery techniques use various responsive materials and stimuli such as an implanted heat source or electronic chip to trigger the drug release from the implanted reservoir. Among many actively controlled stimuli for pulsatile drug delivery, the electrical signal would be the best source because it is portable and does not need large or special equipment to trigger it. The signal can also be easily and on-demand controllable, and long cycles are possible. Furthermore, when a sensor or microchip system is combined, the feedback and remote control outside the body is possible. Researchers have now developed a new drug delivery device for pulsatile and on-demand drug release based on electrically actuatable nanoporous membranes made of polypyrrole.
Feb 10th, 2011
 Spinal cord injury in humans remains a devastating and incurable disorder. Rapid progress in tissue engineering, especially electrospinning techniques that lead to micro- and nanofibrous flexible tubular scaffolds for nerve cell regeneration, may lead to promising therapies for spinal cord injuries. have now demonstrated the repair of a chronically injured spinal cord by attempting to replace the fluid-filled cyst found in these lesions with a neuroprosthetics conducive to tissue reconstruction and axonal regeneration. They managed, for the first time, to obtain a consistent regeneration of the nervous tissue in chronicized injuries at the spinal cord by using a nanostructured composite scaffold with no cells in it.
Spinal cord injury in humans remains a devastating and incurable disorder. Rapid progress in tissue engineering, especially electrospinning techniques that lead to micro- and nanofibrous flexible tubular scaffolds for nerve cell regeneration, may lead to promising therapies for spinal cord injuries. have now demonstrated the repair of a chronically injured spinal cord by attempting to replace the fluid-filled cyst found in these lesions with a neuroprosthetics conducive to tissue reconstruction and axonal regeneration. They managed, for the first time, to obtain a consistent regeneration of the nervous tissue in chronicized injuries at the spinal cord by using a nanostructured composite scaffold with no cells in it.
Feb 2nd, 2011
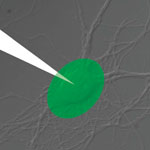 One area of nanobiotechnology that will have a massive impact on improving the live of disabled people lies at the interface (literally) between artificial functional materials and living neuronal tissues. Neuroprosthetics is an area of neuroscience that uses artificial microdevices to replace the function of impaired nervous systems or sensory organs. Different biomedical devices implanted in the central nervous system, so-called neural interfaces, already have been developed to control motor disorders or to translate willful brain processes into specific actions by the control of external devices. One are that has been quite challenging is the communication between biological tissues and artificial sensors - something that is key in building artificial retinas, for instance. Researchers in Italy have now reported the functional interfacing of an organic semiconductor with a network of cultured primary neurons.
One area of nanobiotechnology that will have a massive impact on improving the live of disabled people lies at the interface (literally) between artificial functional materials and living neuronal tissues. Neuroprosthetics is an area of neuroscience that uses artificial microdevices to replace the function of impaired nervous systems or sensory organs. Different biomedical devices implanted in the central nervous system, so-called neural interfaces, already have been developed to control motor disorders or to translate willful brain processes into specific actions by the control of external devices. One are that has been quite challenging is the communication between biological tissues and artificial sensors - something that is key in building artificial retinas, for instance. Researchers in Italy have now reported the functional interfacing of an organic semiconductor with a network of cultured primary neurons.
Jan 26th, 2011
 Brain-computer interfaces, neural probes, brain implants - they all require intensive in vivo studies on how to best combine inorganic electronics with organic neurons. Currently, most neural culture studies suffer from the fact that their cells migrate on a flat surface and are directly exposed to the culture solution, which do not reflect the microenvironment in vivo - neurons, though, can alter their behaviors dramatically in response to the environment change. Ideal cultures, therefore, should mimic the native neural microenvironment to capture the normal cell behavior. This has motivated a group of researchers to come up with this a semiconductor nanomembrane tube approach, which provides a 3D confinement that can potentially isolate the cells from the culture solution. Since these tubes are made from a semiconductor material, it means that they can be integrated with electronic functionalities such as voltage sensors.
Brain-computer interfaces, neural probes, brain implants - they all require intensive in vivo studies on how to best combine inorganic electronics with organic neurons. Currently, most neural culture studies suffer from the fact that their cells migrate on a flat surface and are directly exposed to the culture solution, which do not reflect the microenvironment in vivo - neurons, though, can alter their behaviors dramatically in response to the environment change. Ideal cultures, therefore, should mimic the native neural microenvironment to capture the normal cell behavior. This has motivated a group of researchers to come up with this a semiconductor nanomembrane tube approach, which provides a 3D confinement that can potentially isolate the cells from the culture solution. Since these tubes are made from a semiconductor material, it means that they can be integrated with electronic functionalities such as voltage sensors.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





