Showing Spotlights 353 - 360 of 544 in category All (newest first):
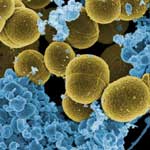 Life-threatening infectious diseases caused by antibiotic-resistant pathogens have been of great concern in both community and hospital settings. This increasing emergence of antibiotic-resistant strains of pathogens has necessitated the development of new antimicrobial surfaces and coatings. As antimicrobial surfaces have become popular in such areas as consumer products, public spaces such as schools and offices, and public transportation, the market for these coatings has quickly grown into a market worth hundreds of million of dollars. New work, by a team from Rensselaer Polytechnic Institute (RPI) has now combined the antimicrobial property of a cell lytic enzyme (lysostaphin) and the excellent properties of carbon nanotubes as an immobilization support in preparing nanocomposite paints that are highly effective against antibiotic-resistant strains of Staphylococcus aureus - methicillin-resistant Staphylococcus aureus (MRSA).
Life-threatening infectious diseases caused by antibiotic-resistant pathogens have been of great concern in both community and hospital settings. This increasing emergence of antibiotic-resistant strains of pathogens has necessitated the development of new antimicrobial surfaces and coatings. As antimicrobial surfaces have become popular in such areas as consumer products, public spaces such as schools and offices, and public transportation, the market for these coatings has quickly grown into a market worth hundreds of million of dollars. New work, by a team from Rensselaer Polytechnic Institute (RPI) has now combined the antimicrobial property of a cell lytic enzyme (lysostaphin) and the excellent properties of carbon nanotubes as an immobilization support in preparing nanocomposite paints that are highly effective against antibiotic-resistant strains of Staphylococcus aureus - methicillin-resistant Staphylococcus aureus (MRSA).
Jul 12th, 2010
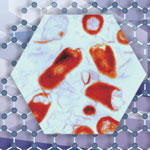 Researchers have made the surprising finding that graphene-based nanomaterials possess excellent antibacterial properties. Although antibacterial materials are widely used in daily life, and the antibacterial properties of nanomaterials are increasingly being explored and developed as commercial products, their cytotoxicity and biocompatibility has raised questions and concerns. Chinese researchers now found that graphene derivatives - graphene oxide, graphene oxide and reduced graphene oxide - can effectively inhibit bacterial growth. This is a significant finding as previous have proven that graphene, particularly graphene oxide, is biocompatible and cells can grow well on graphene substrates. Furthermore, while silver and silver nanoparticles have been well know to be antibacterial, they and other nanomaterials are often cytotoxic.
Researchers have made the surprising finding that graphene-based nanomaterials possess excellent antibacterial properties. Although antibacterial materials are widely used in daily life, and the antibacterial properties of nanomaterials are increasingly being explored and developed as commercial products, their cytotoxicity and biocompatibility has raised questions and concerns. Chinese researchers now found that graphene derivatives - graphene oxide, graphene oxide and reduced graphene oxide - can effectively inhibit bacterial growth. This is a significant finding as previous have proven that graphene, particularly graphene oxide, is biocompatible and cells can grow well on graphene substrates. Furthermore, while silver and silver nanoparticles have been well know to be antibacterial, they and other nanomaterials are often cytotoxic.
Jul 5th, 2010
 A U.S.-Japanese research team has now developed methods for synthesizing protein-sized polymer particles with a binding affinity and selectivity comparable to those of natural antibodies by combining molecular imprinting nanoparticle synthesis with a functional monomer optimization strategy. In effect, they have created a plastic antibody, an artificial version of the real thing. They have also demonstrated that it works in the bloodstream of a living animal. As a result, we can now consider synthetic polymer nanoparticles, prepared by an abiotic process in the chemical laboratory, as alternatives to biological macromolecules. Applications could include antidotes for toxins, protein purification and therapies that currently use antibodies.
A U.S.-Japanese research team has now developed methods for synthesizing protein-sized polymer particles with a binding affinity and selectivity comparable to those of natural antibodies by combining molecular imprinting nanoparticle synthesis with a functional monomer optimization strategy. In effect, they have created a plastic antibody, an artificial version of the real thing. They have also demonstrated that it works in the bloodstream of a living animal. As a result, we can now consider synthetic polymer nanoparticles, prepared by an abiotic process in the chemical laboratory, as alternatives to biological macromolecules. Applications could include antidotes for toxins, protein purification and therapies that currently use antibodies.
Jun 11th, 2010
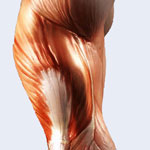 The development of artificial muscles is one of the key areas for bionic enhancements or replacements. The discovery of the electromechanical actuation properties of single-walled carbon nanotubes and the complex behavior of multi-walled carbon nanotubes has led to the development of various carbon nanotube actuators. Besides artificial muscles, potential applications include microelectro-mechanical systems (MEMS), biomimetic micro-and nanorobots, and micro fluidic devices. Recently, a new class of active system, carbon nanotube/polymer composite actuators, has received great attention with regard to macroscopic artificial muscle applications. It has been demonstrated that successful introduction of the highly conductive CNTs could significantly enhance the polymer nanocomposite's electrical, thermal, mechanical, and interface properties, thus providing a suitable material for novel artificial muscle-like actuator investigations.
The development of artificial muscles is one of the key areas for bionic enhancements or replacements. The discovery of the electromechanical actuation properties of single-walled carbon nanotubes and the complex behavior of multi-walled carbon nanotubes has led to the development of various carbon nanotube actuators. Besides artificial muscles, potential applications include microelectro-mechanical systems (MEMS), biomimetic micro-and nanorobots, and micro fluidic devices. Recently, a new class of active system, carbon nanotube/polymer composite actuators, has received great attention with regard to macroscopic artificial muscle applications. It has been demonstrated that successful introduction of the highly conductive CNTs could significantly enhance the polymer nanocomposite's electrical, thermal, mechanical, and interface properties, thus providing a suitable material for novel artificial muscle-like actuator investigations.
May 19th, 2010
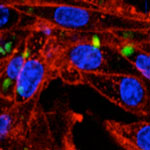 Cultured mammalian cells prefer growing on structured rather than on completely flat surfaces. In regenerative medicine, in which human cells must grow on artificial scaffolds to replace damaged tissue, appropriate biological signals, such as growth factors, but also mechanic stimuli should be provided at the nano and microscales for cell attachment and proliferation, mimicking the natural cell matrices in organic tissues. The straightforward fabrication of nanostructured surfaces as scaffolds for tissue engineering is complex, but instead, micro- and nanorugosities can be easily generated on flat surfaces by either top-down or bottom-up approaches. Researchers have demonstrated that bacterial inclusion bodies formed by biologically irrelevant polypeptides are convenient biomaterials for the bottom-up decoration of substrates for mammalian cell growth.
Cultured mammalian cells prefer growing on structured rather than on completely flat surfaces. In regenerative medicine, in which human cells must grow on artificial scaffolds to replace damaged tissue, appropriate biological signals, such as growth factors, but also mechanic stimuli should be provided at the nano and microscales for cell attachment and proliferation, mimicking the natural cell matrices in organic tissues. The straightforward fabrication of nanostructured surfaces as scaffolds for tissue engineering is complex, but instead, micro- and nanorugosities can be easily generated on flat surfaces by either top-down or bottom-up approaches. Researchers have demonstrated that bacterial inclusion bodies formed by biologically irrelevant polypeptides are convenient biomaterials for the bottom-up decoration of substrates for mammalian cell growth.
May 7th, 2010
 Hydrogels have been in development for several decades and are being used as parts in biology-based microdevices and medical diagnostic technologies, for drug delivery, and in tissue engineering. These gels are networks of water-insoluble polymer chains that are attractive for tissue engineering since their physical and biological properties can be tailored to mimic tissues. Researchers have found that the encapsulation of cells within cell-laden microgels is an attractive approach for engineered tissue formation. Scientists have now developed a technique for the self-assembly of cell-laden microgels on the interface of air and hydrophobic solutions to fabricate three-dimensional tissue constructs with controllable microscale features.
Hydrogels have been in development for several decades and are being used as parts in biology-based microdevices and medical diagnostic technologies, for drug delivery, and in tissue engineering. These gels are networks of water-insoluble polymer chains that are attractive for tissue engineering since their physical and biological properties can be tailored to mimic tissues. Researchers have found that the encapsulation of cells within cell-laden microgels is an attractive approach for engineered tissue formation. Scientists have now developed a technique for the self-assembly of cell-laden microgels on the interface of air and hydrophobic solutions to fabricate three-dimensional tissue constructs with controllable microscale features.
Apr 28th, 2010
 If you think that building an artificial human brain is science fiction, you are probably right - for now. But don't think for a moment that researchers are not working hard on laying the foundations for what is called neuromorphic engineering - a new interdisciplinary discipline that includes nanotechnologies and whose goal is to design artificial neural systems with physical architectures similar to biological nervous systems. One of the key components of any neuromorphic effort is the design of artificial synapses. The human brain contains vastly more synapses than neurons - by a factor of about 10,000 - and therefore it is necessary to develop a nanoscale, low power, synapse-like device if scientists want to scale neuromorphic circuits towards the human brain level. New research now suggests that memristor devices are capable to emulate the biological synapses with properly designed CMOS neuron components.
If you think that building an artificial human brain is science fiction, you are probably right - for now. But don't think for a moment that researchers are not working hard on laying the foundations for what is called neuromorphic engineering - a new interdisciplinary discipline that includes nanotechnologies and whose goal is to design artificial neural systems with physical architectures similar to biological nervous systems. One of the key components of any neuromorphic effort is the design of artificial synapses. The human brain contains vastly more synapses than neurons - by a factor of about 10,000 - and therefore it is necessary to develop a nanoscale, low power, synapse-like device if scientists want to scale neuromorphic circuits towards the human brain level. New research now suggests that memristor devices are capable to emulate the biological synapses with properly designed CMOS neuron components.
Apr 23rd, 2010
 Mass culture of cell lines has long been fundamental to the manufacture of viral vaccines and many products of biotechnology. More recently it also has become an essential tool in stem cell research and tissue engineering. Most conventional petri dish based cell culture techniques produce monolayers cell growth that is missing essential cellular functions that are present in living organisms; gene expression, signaling and morphology can be different and this compromises its clinical relevance. It limits their potential to predict the cellular responses of real organisms. In order to develop cellular models that mimic the functions of living tissues, researchers have therefore been trying to move from two-dimensional to three-dimensional cultures. A recent example is a technique that uses magnetic levitation of cells in the presence of a hydrogel consisting of gold, magnetic iron oxide nanoparticles and filamentous bacteriophage. By spatially controlling the magnetic field while cells divide and grow, the geometry of the cell mass can be manipulated, and multicellular clustering of different cell types in co-culture can be achieved.
Mass culture of cell lines has long been fundamental to the manufacture of viral vaccines and many products of biotechnology. More recently it also has become an essential tool in stem cell research and tissue engineering. Most conventional petri dish based cell culture techniques produce monolayers cell growth that is missing essential cellular functions that are present in living organisms; gene expression, signaling and morphology can be different and this compromises its clinical relevance. It limits their potential to predict the cellular responses of real organisms. In order to develop cellular models that mimic the functions of living tissues, researchers have therefore been trying to move from two-dimensional to three-dimensional cultures. A recent example is a technique that uses magnetic levitation of cells in the presence of a hydrogel consisting of gold, magnetic iron oxide nanoparticles and filamentous bacteriophage. By spatially controlling the magnetic field while cells divide and grow, the geometry of the cell mass can be manipulated, and multicellular clustering of different cell types in co-culture can be achieved.
Mar 31st, 2010
 Life-threatening infectious diseases caused by antibiotic-resistant pathogens have been of great concern in both community and hospital settings. This increasing emergence of antibiotic-resistant strains of pathogens has necessitated the development of new antimicrobial surfaces and coatings. As antimicrobial surfaces have become popular in such areas as consumer products, public spaces such as schools and offices, and public transportation, the market for these coatings has quickly grown into a market worth hundreds of million of dollars. New work, by a team from Rensselaer Polytechnic Institute (RPI) has now combined the antimicrobial property of a cell lytic enzyme (lysostaphin) and the excellent properties of carbon nanotubes as an immobilization support in preparing nanocomposite paints that are highly effective against antibiotic-resistant strains of Staphylococcus aureus - methicillin-resistant Staphylococcus aureus (MRSA).
Life-threatening infectious diseases caused by antibiotic-resistant pathogens have been of great concern in both community and hospital settings. This increasing emergence of antibiotic-resistant strains of pathogens has necessitated the development of new antimicrobial surfaces and coatings. As antimicrobial surfaces have become popular in such areas as consumer products, public spaces such as schools and offices, and public transportation, the market for these coatings has quickly grown into a market worth hundreds of million of dollars. New work, by a team from Rensselaer Polytechnic Institute (RPI) has now combined the antimicrobial property of a cell lytic enzyme (lysostaphin) and the excellent properties of carbon nanotubes as an immobilization support in preparing nanocomposite paints that are highly effective against antibiotic-resistant strains of Staphylococcus aureus - methicillin-resistant Staphylococcus aureus (MRSA).
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





