Showing Spotlights 393 - 400 of 544 in category All (newest first):
 Millions of people with high cholesterol levels are treated with anti-hypolipidemic drugs based on statins that are commonly used to inhibit cholesterol synthesis and lower its serum level. Unfortunately, statins can have two major side effects, although they occur relatively rarely: raised liver enzymes and skeletal muscle pain or even damage. Pharmaceutical research efforts are therefore underway to develop alternative compounds that avoid these potential problems. A promising drug that works via a different mechanism than statin-based drugs, Probucol (PBC), has several advantages over other drugs - better acceptance, ease of administration, and it is much cheaper. Its downside is that its solubility is extremely poor, which considerably lowers its efficiency to suppress cholesterol. A Japanese-U.S. team has now shown that a nanoparticle processing approach enhances the bioavailability of PBC and they demonstrate the design of a solid dosage form for practical use.
Millions of people with high cholesterol levels are treated with anti-hypolipidemic drugs based on statins that are commonly used to inhibit cholesterol synthesis and lower its serum level. Unfortunately, statins can have two major side effects, although they occur relatively rarely: raised liver enzymes and skeletal muscle pain or even damage. Pharmaceutical research efforts are therefore underway to develop alternative compounds that avoid these potential problems. A promising drug that works via a different mechanism than statin-based drugs, Probucol (PBC), has several advantages over other drugs - better acceptance, ease of administration, and it is much cheaper. Its downside is that its solubility is extremely poor, which considerably lowers its efficiency to suppress cholesterol. A Japanese-U.S. team has now shown that a nanoparticle processing approach enhances the bioavailability of PBC and they demonstrate the design of a solid dosage form for practical use.
Mar 31st, 2009
 Carbon nanotubes (CNTs) have already been explored as drug carriers into mammalian cells. Compared to nanoparticles, CNTs have a larger inner volume which allows more drug molecules to be encapsulated, and this volume is more easily accessible because the end caps can be easily removed, and they have distinct inner and outer surfaces for functionalization. In addition to nanomedicine applications, plant science research focusing on investigation of plant genomics and gene function as well as improvement of crop species has become a nanotechnology frontier. To what degree nanomaterials can be employed in delivering payloads into plant cells is a subject that has not yet been explored very well although there appears to be demand from plant cell biologists to take advantage of nanomaterials.
Carbon nanotubes (CNTs) have already been explored as drug carriers into mammalian cells. Compared to nanoparticles, CNTs have a larger inner volume which allows more drug molecules to be encapsulated, and this volume is more easily accessible because the end caps can be easily removed, and they have distinct inner and outer surfaces for functionalization. In addition to nanomedicine applications, plant science research focusing on investigation of plant genomics and gene function as well as improvement of crop species has become a nanotechnology frontier. To what degree nanomaterials can be employed in delivering payloads into plant cells is a subject that has not yet been explored very well although there appears to be demand from plant cell biologists to take advantage of nanomaterials.
Mar 27th, 2009
 The future of tissue and cell engineering depends on the development of next-generation biomaterials that have full control over cell attachment and development into tissue. Since surface topography influences many aspects of cellular and molecular responses, surfaces of implanted devices for instance will one day be engineered to the desired cell shape and cell responses at the point of implantation. The usual techniques of cell patterning are based on passive methods where the intrinsic adhesive properties of the cell are exploited. By creating substrates presenting different areas with particular adhesive characteristics, one can segregate cells on the substrate plane. The main drawback of these techniques is their irreversibility since the differential adhesiveness is permanent. Researchers in France have investigated a new direction for three-dimensional cell patterning that could find applications in tissue engineering. Rather than relying on substrate chemical or physical modifications, they perform the cell patterning using external magnetic forces with which they control the organization of cells on a substrate and create a 3D multicellular assembly.
The future of tissue and cell engineering depends on the development of next-generation biomaterials that have full control over cell attachment and development into tissue. Since surface topography influences many aspects of cellular and molecular responses, surfaces of implanted devices for instance will one day be engineered to the desired cell shape and cell responses at the point of implantation. The usual techniques of cell patterning are based on passive methods where the intrinsic adhesive properties of the cell are exploited. By creating substrates presenting different areas with particular adhesive characteristics, one can segregate cells on the substrate plane. The main drawback of these techniques is their irreversibility since the differential adhesiveness is permanent. Researchers in France have investigated a new direction for three-dimensional cell patterning that could find applications in tissue engineering. Rather than relying on substrate chemical or physical modifications, they perform the cell patterning using external magnetic forces with which they control the organization of cells on a substrate and create a 3D multicellular assembly.
Mar 26th, 2009
 Ever since doctors started replacing worn or damaged bones and teeth with plastic, metal, or ceramic parts, scientists have been on a quest to develop the perfect material for these orthopedic and dental implants. Initially, the challenge was to overcome the body's response to foreign materials, i.e. the strong tendency to reject them. While a lot of progress has been made, and millions of patients receive implants every year ranging from teeth to hip joints, medical implants still do not achieve the same fit and stability as the original tissue that they replace. Researchers have found that the response of host organisms to nanomaterials is different than that observed to conventional materials and that nanopatterning of the surface of implant materials therefore leads to much more compatible prostheses. One approach to improving the biological performance of implants is by functionalizing a non-physiological metallic implant surface through the application of biologically active coatings. Researchers in The Netherlands are now proposing a simple and cost-effective alternative to traditional biomedical coatings for bone implants.
Ever since doctors started replacing worn or damaged bones and teeth with plastic, metal, or ceramic parts, scientists have been on a quest to develop the perfect material for these orthopedic and dental implants. Initially, the challenge was to overcome the body's response to foreign materials, i.e. the strong tendency to reject them. While a lot of progress has been made, and millions of patients receive implants every year ranging from teeth to hip joints, medical implants still do not achieve the same fit and stability as the original tissue that they replace. Researchers have found that the response of host organisms to nanomaterials is different than that observed to conventional materials and that nanopatterning of the surface of implant materials therefore leads to much more compatible prostheses. One approach to improving the biological performance of implants is by functionalizing a non-physiological metallic implant surface through the application of biologically active coatings. Researchers in The Netherlands are now proposing a simple and cost-effective alternative to traditional biomedical coatings for bone implants.
Mar 18th, 2009
 In previous Spotlights we have addressed the numerous benefits that nanotechnology materials and applications could bring to the field of neural engineering and neural prostheses. Different biomedical devices implanted in the central nervous system, so-called neural interfaces, already have been developed to control motor disorders or to translate willful brain processes into specific actions by the control of external devices. Examples of existing brain implants include brain pacemakers, to ease the symptoms of such diseases as epilepsy, Parkinson's Disease, dystonia and recently depression; retinal implants that consist of an array of electrodes implanted on the back of the retina, a digital camera worn on the user's body, and a transmitter/image processor that converts the image to electrical signals sent to the brain. As promising as these new devices are, the reliability and robustness of neural interfaces is a major challenge due to the way brain tissue responds to the implant.
In previous Spotlights we have addressed the numerous benefits that nanotechnology materials and applications could bring to the field of neural engineering and neural prostheses. Different biomedical devices implanted in the central nervous system, so-called neural interfaces, already have been developed to control motor disorders or to translate willful brain processes into specific actions by the control of external devices. Examples of existing brain implants include brain pacemakers, to ease the symptoms of such diseases as epilepsy, Parkinson's Disease, dystonia and recently depression; retinal implants that consist of an array of electrodes implanted on the back of the retina, a digital camera worn on the user's body, and a transmitter/image processor that converts the image to electrical signals sent to the brain. As promising as these new devices are, the reliability and robustness of neural interfaces is a major challenge due to the way brain tissue responds to the implant.
Mar 12th, 2009
 In the quest to make bone, joint and tooth implants almost as good as nature's own version, scientists are turning to nanotechnology. Researchers have found that the response of host organisms to nanomaterials is different than that observed to conventional materials. While this new field of nanomedical implants is in its very early stage, it holds the promise of novel and improved implant materials. One recent example is the nanopatterning of metal surfaces that promises to lead to superior medical implants. A multidisciplinary team of scientists have demonstrated that a simple and inexpensive chemical treatment can create nanopatterns on the surface of different implantable metals, such as Titanium, Tantalum, and CrCoMo alloys.
In the quest to make bone, joint and tooth implants almost as good as nature's own version, scientists are turning to nanotechnology. Researchers have found that the response of host organisms to nanomaterials is different than that observed to conventional materials. While this new field of nanomedical implants is in its very early stage, it holds the promise of novel and improved implant materials. One recent example is the nanopatterning of metal surfaces that promises to lead to superior medical implants. A multidisciplinary team of scientists have demonstrated that a simple and inexpensive chemical treatment can create nanopatterns on the surface of different implantable metals, such as Titanium, Tantalum, and CrCoMo alloys.
Feb 26th, 2009
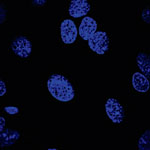 One of the many application areas that carbon nanotubes (CNTs) are experimented with is as a drug carrier in nanomedicine. Typically, nanoparticles have been used for drug delivery and it is only recently that carbon nanotubes have gained attention as potential drug delivery vehicles. Current research has shown the ability of CNTs to carry a variety of molecules such as drugs, DNA, proteins, peptides, targeting ligands etc. into cells - which makes them suitable candidates for targeted delivery applications. Polyethylene glycol (PEG) with molecular weight between 1 and 40 kDa is usually used to coat drug or imaging nanocarriers with the purpose of reducing non-specific interactions with cells and thus increasing the nanocarriers circulation time in vivo. However, when PEG in the form of PL-PEG (adsorbing phospholipid-PEG) has been used to functionalize single-walled CNTs, the finding were not consistent, sometimes even contradictory.
One of the many application areas that carbon nanotubes (CNTs) are experimented with is as a drug carrier in nanomedicine. Typically, nanoparticles have been used for drug delivery and it is only recently that carbon nanotubes have gained attention as potential drug delivery vehicles. Current research has shown the ability of CNTs to carry a variety of molecules such as drugs, DNA, proteins, peptides, targeting ligands etc. into cells - which makes them suitable candidates for targeted delivery applications. Polyethylene glycol (PEG) with molecular weight between 1 and 40 kDa is usually used to coat drug or imaging nanocarriers with the purpose of reducing non-specific interactions with cells and thus increasing the nanocarriers circulation time in vivo. However, when PEG in the form of PL-PEG (adsorbing phospholipid-PEG) has been used to functionalize single-walled CNTs, the finding were not consistent, sometimes even contradictory.
Feb 16th, 2009
 In a previous Nanotechnology Spotlight, we describe how, in order to develop tomorrow's supermaterials, scientists need to unlock nature's structural design rules, in particular for nanoscopic hierarchical molecular structures, and make them available to engineers. This is only possible through a deep understanding of the structure-property relations in biological materials. There is also a surprising relationship between these material design issues and the understanding (or rather lack thereof) of genetic diseases, where structural changes are due to mutations on the molecular level that lead to changed chemical and mechanical properties, which in turn lead to a malfunction of the protein network under mechanical load. Hierarchical nanostructures - ranging through atomistic, molecular and macroscopic scales - represent universal features of biological protein materials. New work by MIT professor Markus Buehler discusses the role of these structural hierarchies in determining properties of biological materials.
In a previous Nanotechnology Spotlight, we describe how, in order to develop tomorrow's supermaterials, scientists need to unlock nature's structural design rules, in particular for nanoscopic hierarchical molecular structures, and make them available to engineers. This is only possible through a deep understanding of the structure-property relations in biological materials. There is also a surprising relationship between these material design issues and the understanding (or rather lack thereof) of genetic diseases, where structural changes are due to mutations on the molecular level that lead to changed chemical and mechanical properties, which in turn lead to a malfunction of the protein network under mechanical load. Hierarchical nanostructures - ranging through atomistic, molecular and macroscopic scales - represent universal features of biological protein materials. New work by MIT professor Markus Buehler discusses the role of these structural hierarchies in determining properties of biological materials.
Jan 27th, 2009
 Millions of people with high cholesterol levels are treated with anti-hypolipidemic drugs based on statins that are commonly used to inhibit cholesterol synthesis and lower its serum level. Unfortunately, statins can have two major side effects, although they occur relatively rarely: raised liver enzymes and skeletal muscle pain or even damage. Pharmaceutical research efforts are therefore underway to develop alternative compounds that avoid these potential problems. A promising drug that works via a different mechanism than statin-based drugs, Probucol (PBC), has several advantages over other drugs - better acceptance, ease of administration, and it is much cheaper. Its downside is that its solubility is extremely poor, which considerably lowers its efficiency to suppress cholesterol. A Japanese-U.S. team has now shown that a nanoparticle processing approach enhances the bioavailability of PBC and they demonstrate the design of a solid dosage form for practical use.
Millions of people with high cholesterol levels are treated with anti-hypolipidemic drugs based on statins that are commonly used to inhibit cholesterol synthesis and lower its serum level. Unfortunately, statins can have two major side effects, although they occur relatively rarely: raised liver enzymes and skeletal muscle pain or even damage. Pharmaceutical research efforts are therefore underway to develop alternative compounds that avoid these potential problems. A promising drug that works via a different mechanism than statin-based drugs, Probucol (PBC), has several advantages over other drugs - better acceptance, ease of administration, and it is much cheaper. Its downside is that its solubility is extremely poor, which considerably lowers its efficiency to suppress cholesterol. A Japanese-U.S. team has now shown that a nanoparticle processing approach enhances the bioavailability of PBC and they demonstrate the design of a solid dosage form for practical use.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





