| Posted: May 18, 2016 |
Fine tuning phosphorous heterocycle materials for organic electronics
(Nanowerk News) Scientists at Tokyo Institute of Technology have produced airstable 1-aryl 1,3-diphosphacyclobutane-2,4-diyl materials by direct arylation with electron rich aromatic substituents ("Access to Air-Stable 1,3-Diphosphacyclobutane-2,4-diyls by an Arylation Reaction with Arynes"). This method that enables fine tuning of the electronic properties of such phosphorous heterocycles compounds for applications including fabrication of organic electronics and hydrogen fluoride sensors.
|
|
Materials scientists have a strong interest in the development of methods for the synthesis of so-called 'open-shell singlet P-heterocyclic' materials systems for applications in the organic electronics industry including organically based sensors and optoelectronic devices. Here, Shigekazu Ito and colleagues at School of Materials and Chemical Technology, Tokyo Institute of Technology report on a new method for the production of airstable 1-aryl 1,3-diphosphacyclobutane-2,4-diyls— an open-shell singlet P-heterocyclic' materials system—by direct arylation with electron rich aromatic substituents.
|
|
This ability to tune the physiochemical nature of the electron-donating 1-aryl 1,3-diphosphacyclobutane-2,4-diyls is expected to find applications including p-type semiconductors and sensors of hydrogen fluoride.
|
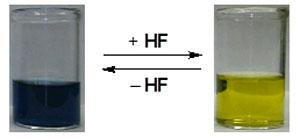 |
| Reversible change of color via the hydrofluorination process.
|
|
Background
|
|
The Tokyo Tech group have previously reported on a nucleophilic aromatic substitution (SNAr) process for producing air-stable 1-aryl 1,3-diphosphacyclobutane-2,4-diyls. However, this method only worked in the case of electron-deficient N-heterocyclic aryl halides were employed as the electrophiles. Hence, there is still the need for new approaches for direct arylation of 1,3-diphosphacyclobuten-4-yl anions with electron rich aromatic substituents that produce electron-donating and stable 1,3-diphosphacyclobutane-2,4-diyls for organic electronics.
|
|
Details
|
|
The method developed by the Tokyo Tech group is based on the assumption that, "highly electrophilic arynes should react with and afford stable and electron-donating openshell singlet P-heterocycles." In their experiments, the researchers generated arynes from the appropriate o-silyl triflates and fluoride, observed them to react with 1,3-diphosphacyclobuten-4-yl anion under appropriate conditions with the result being air-stable open-shell singlet P-heterocycles.
|
|
Notably, during the experiments the researchers also detected the presence of hydrogen fluoride (HF) by the P-arylated 1,3 diphosphacyclobutane-2,4-diyls. This release of HF resulted in remarkable changes in their photoabsorption properties. Analysis showed that 1,3 diphosphacyclobutane-2,4-diyls absorbed visible light at approximately 600 nm. Futhermore, the researchers fabricated a field effect transistor with a carrier mobility of 1.29x 108 cm2/ Vs, ON/OFF ratio of 6, and threshold voltage of 4 V.
|
|
The Tokyo Tech group also found the optical absorption to show a 'blue shift' when the open-shell P-heterocyclic system 'trapped' hydrogen fluoride (HF); a finding that may enable the visual detection of the existence of HF, and could be used for the development of HF sensors.
|
|
The researchers state: "In our study of HF trapping by the open-shell P-heterocyclic system, the electron-rich 1,3-diphosphacyclobutane-2,4-diyl system showed a remarkable blueshift of photoabsorption assigned as the transition from the HOMO to the LUMO, which enabled the identification of the presence of HF by visual observation."
|

