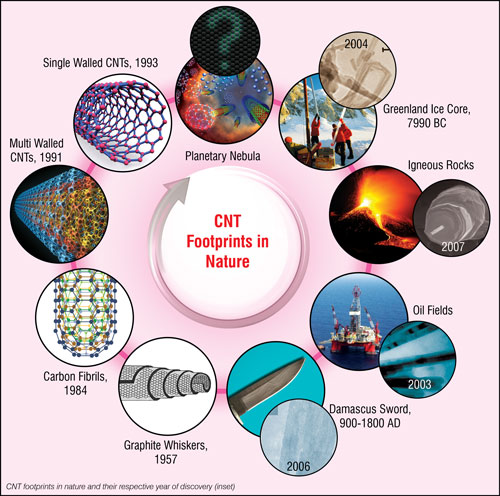
Carbon nanotubes – what they are, how they are made, what they are used for
Content
What are carbon nanotubes?
Carbon nanotubes (CNTs) are cylindrical molecules that consist of rolled-up sheets of single-layer carbon atoms (graphene). They can be single-walled (SWCNT) with a diameter of less than 1 nanometer (nm) or multi-walled (MWCNT), consisting of several concentrically interlinked nanotubes, with diameters reaching more than 100 nm. Their length can reach several micrometers or even millimeters.

The purple structure is a human hair fragment, with a diameter of about 80 to 100 thousand nanometers and in the background is a network of single-walled carbon nanotubes. (Image: Jirka Cech)
Like their building block graphene (Why not read our extensive tutorial on graphene – which includes a fantastic infographic), CNTs are chemically bonded with sp2 bonds, an extremely strong form of molecular interaction.
This feature combined with carbon nanotubes' natural inclination to rope together via van der Waals forces, provide the opportunity to develop ultra-high strength, low-weight materials that possess highly conductive electrical and thermal properties. This makes them highly attractive for numerous applications.

Carbon allotropes
Carbon is the fourth-most-abundant element in the universe and, depending on the arrangements of carbon atoms, takes on a wide variety of forms, called allotropes. Carbon allotropes exhibit unique properties of strength and electrical conductivity.
Solid carbon at room temperature has two classical structures: diamond and graphite. In 1985 the discovery of the existence of a third and new carbon allotrope containing sixty perfectly symmetrically arranged carbon atoms (also known as C60, fullerene, or buckyballs) meant a major breakthrough and opened a novel field of carbon nanochemistry. Then, in 1991, carbon nanotubes were discovered and graphene in 2004.
Electrical properties of carbon nanotubes
The rolling-up direction (rolling-up or chiral vector) of the graphene layers determines the electrical properties of the nanotubes. Chirality describes the angle of the nanotube's hexagonal carbon-atom lattice.
Armchair nanotubes – so called because of the armchair-like shape of their edges – have identical chiral indices and are highly desired for their perfect conductivity. They are unlike zigzag nanotubes, which may be semiconductors. Turning a graphene sheet a mere 30 degrees will change the nanotube it forms from armchair to zigzag or vice versa.
While MWCNTs are always conducting and achieve at least the same level of conductivity as metals, SWCNTs' conductivity depends on their chiral vector: they can behave like a metal and be electrically conducting; display the properties of a semi-conductor; or be non-conducting. For example, a slight change in the pitch of the helicity can transform the tube from a metal into a large-gap semiconductor.
Properties of carbon nanotubes
Apart from their electrical properties, which they inherit from graphene, CNTs also have unique thermal and mechanical properties that make them intriguing for the development of new materials:
All these properties make carbon nanotubes ideal candidates for electronic devices, chemical/electrochemical and biosensors, transistors, electron field emitters, lithium-ion batteries, white light sources, hydrogen storage cells, cathode ray tubes (CRTs), electrostatic discharge (ESD) and electrical-shielding applications.
Difference between carbon nanotubes and carbon nanofibers
Please note that carbon nanotubes are different than carbon nanofibers (CNFs). CNFs are usually several micrometers long and have a diameter of about 200 nm. Carbon fibers have been used for decades to strengthen compound but they do not have the same lattice structure as CNTs. Instead, they consist of a combination of several forms of carbon and/or several layers of graphite, which are stacked at various angles on amorphous carbon (where atoms do not arrange themselves in ordered structures). CNFs have similar properties as CNTs, but their tensile strength is lower owing to their variable structure and they are not hollow inside.
For starters, you could watch these five short videos about carbon nanotubes:
Who discovered carbon nanotubes?
Thousands of papers are being published every year on CNTs or related areas and most of these papers give credit for the discovery of CNTs to Sumio Iijima who, in 1991, published a ground-breaking paper in Nature ("Helical microtubules of graphitic carbon") reporting the discovery of multi-walled carbon nanotubes.
On taking a cursory look at the scientific literature, one might get the impression that Iijima is the de facto discoverer of carbon nanotubes. Of course, there is no doubt that he has made two seminal contributions to the field, however a careful analysis of the literature suggests that certainly he is not the first one who has reported the existence of CNTs.
An editorial in the journal Carbon ("Who should be given the credit for the discovery of carbon nanotubes?") tried to clear the air by describing the chronological events that led to the discovery of carbon nanotubes. By delving deeper into the history of carbon nanotubes, it becomes quite apparent that the origin of CNTs could be even pre-historic in nature (read more here in our article on the birth and early history of carbon nanotubes.
How are carbon nanotubes made?
Three main methods are currently available for the production of CNTs: arc discharge, laser ablation of graphite, and chemical vapor deposition (CVD).
In the first two processes, graphite is combusted electrically or by means of a laser, and the CNTs developing in the gaseous phase are separated. All three methods require the use of metals (e.g. iron, cobalt, nickel) as catalysts.
CVD process
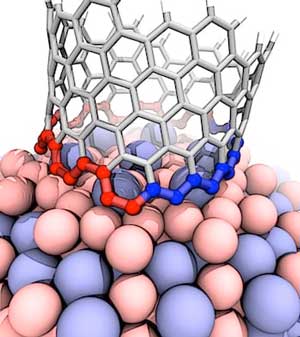
The CVD process currently holds the greatest promise, since it allows the production of larger quantities of CNTs under more easily controllable conditions and at lower cost. In the CVD process, manufacturers can combine a metal catalyst (such as iron) with carbon-containing reaction gases (such as hydrogen or carbon monoxide) to form carbon nanotubes on the catalyst inside a high-temperature furnace.

Schematic view of CNT growth on catalyst particles during CVD. First, small secondary catalyst particles of the size of a CNT diameter develop, on which the nanotubes start growing. The catalyst particle is either at the top or at the bottom of the emerging nanotube. Growth will stop if the catalyst particle is deactivated through the development of a carbon envelope. (Image: © Royal Society of Chemistry)
sporting goods(bicycle frames, tennis rackets, hockey sticks, golf clubs and balls, skis, kayaks; sports arrows)
yachting (masts, hulls and other parts of sailboats)
textiles (antistatic and electrically conducting textiles ('smart textiles'); bullet-proof vests, water-resistant and flame-retardant textiles)
automotive, aeronautics and space (light-weight, high-strength structural composites)
industrial engineering (e.g. coating of wind-turbine rotor blades, industrial robot arms)
electrostatic charge protection (for instance, researchers have a developed electrically conducting and flexible CNT film specifically for space applications) and radiation shielding with CNT-based nanofoams and aerogels.

CNT fabric stopped a 9MM, jacketed round in controlled ballistics testing. This material shown is roughly the same thickness as six stacked business cards. (Source: Nanocomp Technologies)

Positive and negative regulation of carbon nanotube catalysts through encapsulation within macrocycles. (Image: Emilio M. Pérez, IMDEA)

Schematic of a sub-10 nm carbon nanotube transistor configuration. (Image: Aaron Scott, Southern Illinois University)

Example of a flexible hydrogen sensor fabricated with single-walled carbon nanotubes. (Image: Dr. Sun/Argonne)
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC

The CVD process can be purely catalytic or plasma-supported. The latter requires slightly lower temperatures (200-500°C) than the catalytic process (up to 750°C) and aims at producing 'lawn-like' CNT growth.
Purification
Even though synthetic techniques have been improved to obtain high-purity carbon
nanotubes, the formation of byproducts containing impurities such as metal encapsulated nanoparticles, metal particles in the tip of a carbon nanotube, and amorphous carbon has been an unavoidable phenomenon, because the metal nanoparticles are essential for the nanotube growth.
These foreign nanoparticles, as well as structural defects that occurred during synthesis, have the unfortunate implication that they modify the physico-chemical properties of the produced carbon nanotubes.
That's why carbon nanotubes need to be purified with the help of various methods such as acid treatment or ultrasound at the end of the production process.
Applications of carbon nanotubes and their uses
CNTs are well-suited for virtually any application requiring high strength, durability, electrical conductivity, thermal conductivity and lightweight properties compared to conventional materials.
Currently, CNTs are mainly used as additives to synthetics. CNTs are commercially available as a powder, i.e. in a highly tangled-up and agglomerated form. For CNTs to unfold their particular properties they need to be untangled and spread evenly in the substrate.
Another requirement is that CNTs need to be chemically bonded with the substrate, e.g. a plastic material. For that purpose, CNTs are functionalized, i.e. their surface is chemically adapted for optimal incorporation into different materials and for the specific application in question.
Carbon nanotubes can also be spun into fibers, which not only promise interesting possibilities for specialty textiles but may also help realize a particularly utopian project – the space elevator.
Materials
Carbon nanotube enabled nanocomposites have received much attention as a highly attractive alternative to conventional composite materials due to their mechanical, electrical, thermal, barrier and chemical properties such as electrical conductivity, increased tensile strength, improved heat deflection temperature, or flame retardancy.
These materials promise to offer increased wear resistance and breaking strength, antistatic properties as well as weight reduction. For instance, it has been estimated that advanced CNT composites could reduce the weight of aircraft and spacecraft by up to 30%.
These composite materials already find use in

Catalysis
What makes carbon nanotubes so attractive for catalysis is their exceptionally high surface area combined with the ability to attach essentially any chemical species to their sidewalls. Already, CNTs have been used as catalysts in many relevant chemical processes, however, controlling their catalytic activity is not easy.
Initially, carbon nanotubes have been combined with molecules via very strong bonds (covalent bonds) that lead to very stable compounds. Such connection, however, implies a change in the structure of the nanotube and therefore in its properties.
It would be analogous to nailing an advertisement to a post using a thumbtack: the union is strong, but it leaves a hole in both the advertisement and the post. Weak non-covalent forces have also been used, which keep the structure of the nanotubes intact, but typically yield kinetically unstable compounds. The comparison in this case would be to tape the advert to the post. Neither the advertisement nor the post is damaged, but the union is much weaker.
To overcome this issue, researchers already are developing methods for the chemical modification of carbon nanotubes by mechanical bonding, the first example of mechanically interlocked carbon nanotubes (MINTs). This type of compounds is as stable as covalent compounds, but at the same time as respectful of the initial structure as the non-covalent compounds.

Transistors
Despite the rise of graphene and other two-dimensional (2D) materials, semiconducting single-walled carbon nanotubes are still regarded as strong candidates for the next generation of high-performance, ultra-scaled and thin-film transistors as well as for opto-electronic devices to replace silicon electronics (read more: "20 years of nanotube transistors").
One of the crucial questions is if CNT transistors can offer performance advantages over silicon at sub-10 nm lengths.
There have been mixed opinions in the nanoelectronics community regarding whether or not CNT transistors would maintain their impressive performance at extremely scaled lengths. Some argued that the very small effective mass of the carriers would contribute to a tunneling phenomena that would cause the devices to breakdown around 15 nm – an opinion supported by the few theoretical studies that explored nanotube devices at such dimensions.
Meanwhile, others remained convinced that the ultrathin body of single-walled carbon nanotubes – only 1 nm in diameter – would allow for excellent transistor behavior even down to the sub-10 nm range.

So far researchers have achieved only promising experimental results and at this point there remain numerous challenges related to integrating CNT transistors into industrial-scale chip manufacturing.
Sensors
The group of Cees Dekker paved the way for the development of CNT-based electrochemical nanosensors by demonstrating the possibilities of SWCNTs as quantum wires and their effectiveness in the development of field-effect transistors.
Many studies have shown that although CNTs are robust and inert structures, their electrical properties are extremely sensitive to the effects of charge transfer and chemical doping by various molecules.
Most sensors based on CNTs are field effect transistors (FET) – although CNT are robust and inert structures, their electrical properties are extremely sensitive to the effects of charge transfer and chemical doping by various molecules. CNTs-FETs have been widely used to detect gases such as greenhouse gases in environmental applications.
The functionalization of CNTs is important for making them selective to the target analyte. Different types of sensors are based based on molecular recognition interactions between functionalism CNT and target analytes.
For instance, researchers have developed flexible hydrogen sensors using single-walled carbon nanotubes decorated with palladium nanoparticles.

Nano inks
Ink formulations based on CNT dispersions are attractive for printed electronics applications such as transparent electrodes, RFID tags, thin-film transistors, light-emitting devices, and solar cells (read more: "Conductive nanomaterials for printed electronics applications").
Electrodes
Carbon nanotubes have been widely used as electrodes for chemical and biological sensing applications and many other electrochemical studies. With their unique one-dimensional molecular geometry of a large surface area coupled with their excellent electrical properties, CNTs have become important materials for the molecular engineering of electrode surfaces where the development of electrochemical devices with region-specific electron-transfer capabilities is of paramount importance.
Displays
Given their high electrical conductivity, and the incredible sharpness of their tip (the smaller the tips' radius of curvature, the more concentrated the electric field, the higher field emission), carbon nanotubes are considered the most promising material for field emitters and a practical example are CNTs as electron emitters for field emission displays (FED).
Field emission display (FED) technology makes possible a new class of large area, high resolution, low cost flat panel displays. However, FED manufacturing requires CNT to be grown in precise sizes and densities. Height, diameter and tip sharpness affect voltage, while density affects current.
Buckypapers
Buckypapers could find numerous applications: As one of the most thermally conductive materials known, buckypaper could lead to the development of more efficient heat sinks for chips; a more energy-efficient and lighter background illumination material for displays; a protective material for electronic circuits from electromagnetic interference due to its unusually high current-carrying capacity; or switchable surfaces (see: "Nanotechnology paper for switchable surfaces").
Optoelectronic and photonic applications
While individual nanotubes generate discrete fine peaks in optical absorption and emission, macroscopic structures consisting of many CNTs gathered together also demonstrate interesting optical behavior.
For example, a millimeter-long bundle of aligned MWCNTs emits polarized incandescent light by electrical current heating and SWCNT bundles are giving higher brightness emission at lower voltage compared with conventional tungsten filaments.
Nanomedicine and biotechnology
Carbon nanomaterials such as nanotubes or graphene not only are widely researched for their potential uses in industrial applications, they also are of great interest to biomedical engineers working on nanotechnology applications.
There is considerable interest in using CNTs for various biomedical applications. The physical properties of CNTs, such as mechanical strength, electrical conductivity, and optical properties, could be of great value for creating advanced biomaterials.
Carbon nanotubes can also be chemically modified to present specific moieties (e.g., functional groups, molecules, and polymers) to impart properties suited for biological applications, such as increased solubility and biocompatibility, enhanced material compatibility and cellular responsiveness.
Nitrogen-doped carbon nanotubes for instance have been developed for drug delivery applications ("Nanoparticle-corked carbon nanotubes as drug delivery vehicles").
However, the issue of cytotoxicity of CNTs is an area that has already attracted much research interest and has not resulted in a definitive answer yet. Given the inconclusive state of these nanotoxicology studies researchers says that more systematic biological evaluations of CNTs having various chemical and physical properties are warranted in order to determine their precise pharmacokinetics, cytotoxicity, and optimal dosages.
Filtration
High-flow membranes are an important part of future energy-efficient water purification. Already, researchers have demonstrated efficient water transport in carbon nanotubes with openings of less than one nanometer.
When embedded in fatty membranes, the nanotubes squeeze entering water molecules into a single file chain, which leads to very fast transport. The flow was 10 times faster than in wider carbon nanotubes and 6 times faster than in the best biological membrane, a protein called aquaporin (read more: "Filtering water better than nature").
Carbon nanotubes also have been used to demonstrate protective textiles with ultra breathable membranes. These membranes provide rates of water vapor transport that surpass those of commercial breathable fabrics like GoreTex, even though the CNT pores are only a few nanometers wide.
Crucially, they also provide protection from biological agents due to their very small pore size, less than 5 nanometers wide. Biological threats like bacteria or viruses are much larger and typically more than 10-nm in size.
In order to make these membranes also protect from chemical agents, which are much smaller in size, researchers modified the CNT surfaces with chemical-threat-responsive functional groups. These functional groups will sense and block the threat like gatekeepers on the pore entrance (read more: "'Second skin' protects soldiers from biological and chemical agents").
Frequently Asked Questions (FAQs) about carbon nanotubes
What are Carbon Nanotubes?
Carbon Nanotubes, or CNTs, are cylindrical nanostructures composed entirely of carbon atoms. These tubes have extraordinary mechanical, electrical, thermal, optical, and chemical properties, which makes them suitable for various applications in nanotechnology, electronics, optics, and other fields of materials science.
How are Carbon Nanotubes Made?
Carbon Nanotubes are generally produced using three methods: Arc Discharge, Laser Ablation, and Chemical Vapor Deposition (CVD). Among these, CVD is the most popular due to its lower cost and higher scalability.
What are the Types of Carbon Nanotubes?
Carbon Nanotubes come in two main types: Single-Walled Carbon Nanotubes (SWCNTs) and Multi-Walled Carbon Nanotubes (MWCNTs). The SWCNTs have a single layer of carbon atoms, while MWCNTs consist of multiple concentric layers of carbon atoms.
What are the Properties of Carbon Nanotubes?
Carbon Nanotubes possess unique properties. They are incredibly lightweight yet stronger than steel, have high thermal conductivity, excellent electrical conductivity, and exhibit unique optical properties. These attributes make them suitable for various technological applications.
What are the Applications of Carbon Nanotubes?
Carbon Nanotubes have diverse applications in many fields, including electronics, materials science, and nanotechnology. They are used in making transistors, batteries, energy storage devices, sensors, and even medical devices. In materials science, they are used for strengthening composite materials.
Are Carbon Nanotubes Safe?
The safety of Carbon Nanotubes is still under investigation. Although they have vast potential for use in various fields, their impact on human health and the environment is not fully understood. Some studies suggest they can be harmful if inhaled, similar to asbestos fibers. Thus, appropriate safety measures should be taken when handling them.
What is the Current Research on Carbon Nanotubes?
Research on Carbon Nanotubes is extensive, focusing on developing new applications, improving production methods, and studying their properties. Additionally, researchers are studying the potential environmental and health impacts of Carbon Nanotubes to ensure their safe usage.
Can Carbon Nanotubes Conduct Electricity?
Yes, Carbon Nanotubes are excellent conductors of electricity. They can carry a high current density without being damaged, making them useful in various electronics applications.
Can Carbon Nanotubes be Used in Medicine?
Yes, Carbon Nanotubes have potential applications in medicine. Their unique properties make them useful for drug delivery, tissue engineering, and as components in medical devices. However, more research is needed to ensure their safety and effectiveness in these applications.
What is the Future of Carbon Nanotubes?
The future of Carbon Nanotubes looks promising, with extensive research exploring their applications in various fields. Their unique properties could revolutionize electronics, medicine, energy storage, and materials science. However, ensuring their safe use is a critical area for future research.
Are Carbon Nanotubes Expensive?
The cost of Carbon Nanotubes can vary greatly depending on their quality, type, and production method. Generally, high-quality Carbon Nanotubes are expensive due to the complexity of their production process. However, as production methods improve, costs may decrease in the future.
How are Carbon Nanotubes Used in Electronics?
In electronics, Carbon Nanotubes are used in transistors, sensors, and other components due to their excellent electrical conductivity and unique electronic properties. They may also play a critical role in developing smaller, faster, and more energy-efficient electronic devices in the future.
What is the Environmental Impact of Carbon Nanotubes?
The environmental impact of Carbon Nanotubes is not fully understood. While they offer significant potential benefits, there are concerns about their potential to harm the environment if not properly disposed of or recycled. Research is ongoing to understand their environmental impact and develop safe handling and disposal methods.
Can Carbon Nanotubes be Recycled?
Research is ongoing to develop methods for recycling Carbon Nanotubes, especially given their potential environmental impact. While some progress has been made, effectively recycling Carbon Nanotubes and maintaining their unique properties remains a significant challenge.
What is the Role of Carbon Nanotubes in Energy Storage?
Carbon Nanotubes have significant potential in energy storage. They are used in supercapacitors and batteries due to their high surface area, electrical conductivity, and chemical stability. They can enhance energy storage devices' performance, making them more efficient and capable of storing more energy.
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC