| Posted: Oct 14, 2014 |
Cheap catalyst gets expensive accessory
|
|
(Nanowerk News) While iron catalysts are an inexpensive way to remove oxygen from plant-based materials, the catalyst is not very active and can be readily deactivated due to rusting or oxidation by the water that comes part and parcel with biofuels production. Precious metal catalysts such as palladium aren't readily oxidized, but they are not efficient in removing oxygen from plant-based materials (low catalyst activity and high hydrogen consumption), on top of the fact that the metal is prohibitively expensive. But adding just a touch of palladium to the iron produces a catalyst that quickly removes oxygen atoms, easily releases the desired products, and doesn't rust, according to scientists at Pacific Northwest National Laboratory (PNNL) and Washington State University (WSU).
|
|
"The synergy between the palladium and the iron is incredible," said Dr. Yong Wang, who led the two papers featured on the cover of ACS Catalysis. "When combined, the catalyst is far better than the metals alone in terms of activity, stability, and selectivity."
|
|
Wang has a joint appointment with PNNL and WSU. At PNNL, he is an associate director at the Institute for Integrated Catalysis; at WSU, he is the Voiland Distinguished Professor.
|
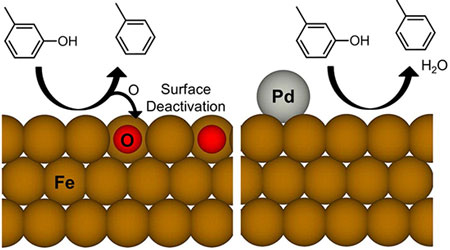 |
| Researchers showed how the addition of palladium (PD) prevents deactivation (addition of oxygen, red spheres) of an iron catalyst in the reaction that removes oxygen from biofuel feedstock. (© American Chemical Society)
|
|
Why It Matters
|
|
To create drop-in biofuels, substitutes for gasoline, diesel fuel, and jet fuel that don't require changing today's fossil fuel based infrastructure, scientists need fast, efficient catalysts to remove oxygen atoms from lignin, which is abundant in plants. High levels of oxygen create energy-poor fuel and would harm systems that deal with fossil fuels. This study shows how a two-metal approach could improve the catalyst and minimize the amount of hydrogen needed, along with the associated costs.
|
|
"With biofuels, you need to remove as much oxygen as possible to gain energy density," said Wang. "Of course, in the process, you want to minimize the costs of oxygen removal. In this case, you minimize hydrogen consumption and increase the overall activity and gain high yields to desired fuel products."
|
|
Methods
|
|
The team combined experimental and theory approaches to determine how the atoms on the catalyst's surface interacted with m-creosol, a model compound of lignin derivatives. The team employed advanced instruments, including high-resolution transmission electron microscopy, X-ray photoelectron spectroscopy, and extended X-ray absorption fine structure spectroscopy. The team also used complex calculations to interpret the experimental view.
|
|
Adding extremely small amounts of palladium to the iron catalyst promoted hydrogen coverage on the iron surface, making the hydrodexoygenation reaction highly favored. The palladium also prevented the iron from being oxidized by water. The palladium further enhanced the release of the desired aromatic molecules. The result: less hydrogen is consumed to remove the oxygen. "By adding the palladium, we could potentially use metals such as iron, which are cheaper and function at better rates with high yields than palladium or iron alone," said Wang.
|
|
What's Next?
|
|
The team is designing catalysts to work under wetter conditions. "Our work involved model compounds; we want to study more realistic conditions where you have more water and you still want to remove as much oxygen as possible," said Wang.
|
|
References
|
|
Hong YC, H Zhang, J Sun, A Karim, A Hensley, M Gu, MH Engelhard, JS McEwen, and Y Wang. 2014. "Synergistic Catalysis between Pd and Fe in Gas Phase Hydrodeoxygenation of m-Cresol". ACS Catalysis
|
|
Hensley AJR, Y Hong, R Zhang, H Zhang, J Sun, Y Wang, and JS McEwen. 2014. "Enhanced Fe2O3 Reducibility via Surface Modification with Pd: Characterizing the Synergy within Pd/Fe Catalysts for Hydrodeoxygenation Reactions" ACS Catalysis.
|

