| Posted: Dec 11, 2014 |
Transforming the composition of nanoparticles using electrochemistry
|
|
(Nanowerk News) Galvanic replacement reactions provide an elegant way of transforming solid nanoparticles into complex hollow morphologies. Conventionally, galvanic replacement is studied by stopping the reaction at different stages and characterizing the products ex situ.
|
|
In situ observations by liquid-cell electron microscopy can provide insight into mechanisms, rates and possible modifications of galvanic replacement reactions in the native solution environment.
|
|
Here, researchers at Brookhaven's Center for Functional Nanomaterials use liquid-cell electron microscopy to investigate galvanic replacement reactions between silver nanoparticle templates and aqueous palladium salt solutions ("In situ liquid-cell electron microscopy of silver–palladium galvanic replacement reactions on silver nanoparticles").
|
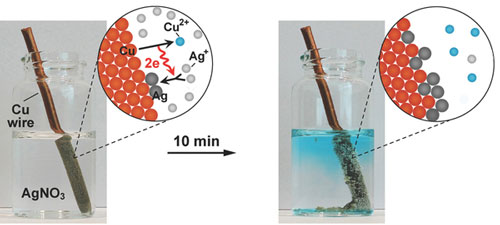 |
| Images of a copper wire undergoing galvanic replacement when immersed in a silver-based solution. The associated cartoons illustrate the replacement mechanism for copper and silver, resulting in silver coating of the wire.
|
|
Their in situ observations follow the transformation of the silver nanoparticles into hollow silver–palladium nanostructures.
|
|
While the silver–palladium nanocages have morphologies similar to those obtained in ex situ control experiments the reaction rates are much higher, indicating that the electron beam strongly affects the galvanic-type process in the liquid-cell.
|
|
By using scavengers added to the aqueous solution the researchers identify the role of radicals generated via radiolysis by high-energy electrons in modifying galvanic reactions.
|
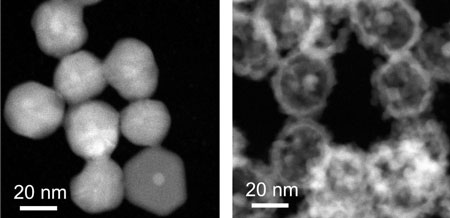 |
| Electron microscopy images of the galvanic replacement reaction, transforming a silver nanoparticle into a hollow silver/palladium nanostructure.
|
|
Why Does This Matter?
|
|
Understanding how to control galvanic replacement reactions in nanoparticles is important for exploiting these materials in applications, such as nanoscale containers for diagnostics and drug delivery, contrast enhancement agents in biomedical imaging, or platforms for facilitating chemical reactions.
|


