| Posted: Mar 20, 2015 | |
Putting batteries on stage spotlights performance at the nanoscale |
|
| (Nanowerk News) Used in everything from electric vehicles to laptop computers, the lithium battery is ubiquitous, but it is not well understood at the atomic scale. To see what happens on the nanoscale, scientists at DOE's Joint Center for Energy Storage Research (JCESR) designed and implemented a small device, known as an operando electrochemical stage. Using this stage inside a state-of-the-art aberration-corrected transmission electron microscope they can take nanoscale-resolution pictures of lithium ions as they are deposited on or dissolve off of an electrode while the battery runs ("Observation and Quantification of Nanoscale Processes in Lithium Batteries by Operando Electrochemical (S)TEM"). | |
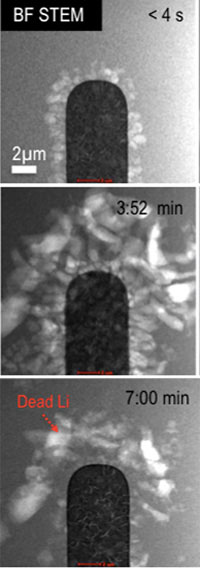 Lithium deposited on the platinum anode at the beginning (top), during (middle) and end (bottom) of the second cycle. Residual “dead lithium” can be seen on and around the anode. With the new stage, scientists can directly image changes as they occur. The new images allow precise measurements and descriptions of what happens inside the battery. This information is vital to control performance- and safety-limiting processes. Now, scientists can rapidly visualize and test new pairings of electrodes and electrolytes (see Battery 101). The new stage will help quickly sort through options for longer lasting, safer batteries. Methods Moving beyond the current industry-standard lithium-ion battery has been difficult. In lithium-air and other designs, interactions at the electrode-electrolyte interfaces affect the battery's performance and safety. To understand the reactions, scientists at the Pacific Northwest National Laboratory, as part of JCESR, created an operando electrochemical stage. Using it in an aberration-corrected scanning transmission electron microscope, scientists can now chemically image the interface between the platinum anode and the electrolyte during the battery operation. The imaging method highlights solid lithium metal, uniquely identifying it from the components that make up the protective solid-electrolyte interphase layer. Using these images and standard electrochemical data, scientists can quantify, at the nanoscale, the amount of lithium that ends up irreversibly deposited after each charge/discharge cycle. This means they can view dendrites -- the microscopic thorns that cause batteries to fail -- as they form. The technique also shows the growth of the solid-electrolyte interphase layer, which wraps around and protects the anode. The layer is formed as a result of the electrolyte breaking down. In their studies, the team found that extended battery cycling leads to lithium growing beneath the layer -- the genesis of the dendrites that have implications for battery safety and performance. |
|
| What's Next? | |
| This new imaging tool opens up possibilities to rapidly visualize and test electrode/electrolyte pairings for new battery systems. These systems could allow electric cars to travel great distances between charges. Also, one day, such systems could store energy from wind and solar stations, making the intermittent energy available when needed. |
| Source: Pacific Northwest National Laboratory |
