| Posted: Mar 26, 2015 |
Gold nanoparticles size up to cancer treatment
|
|
(Nanowerk News) Treatments that attack cancer cells through the targeted silencing of cancer genes could be developed using small interfering RNA molecules (siRNA). However delivering the siRNA into the cells intact is a challenge as it is readily degraded by enzymes in the blood and small enough to be eliminated from the blood stream by kidney filtration.
|
|
Now Kazunori Kataoka at the University of Tokyo and colleagues at Tokyo Institute of Technology have designed a protective treatment delivery vehicle with optimum stability and size for delivering siRNA to cells (ACS Nano, "Precise engineering of siRNA delivery vehicles to tumors using polyion complexes and gold nanoparticles").
|
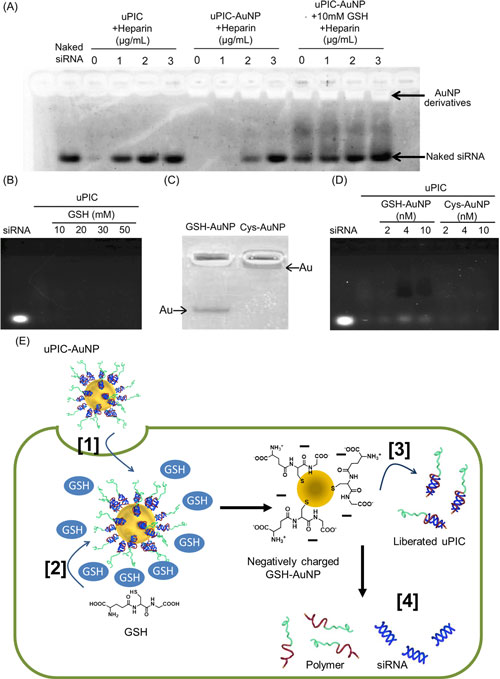 |
| Top: Stability assay of the siRNA-loaded polymer complex (labelled uPIC, unimer polyion complex) without and with gold nanoparticles (AuNP) incubated with heparin at 0, 1, 2, and 3 µg/mL, and glutathione (GSH) at 0 and 10 mM. Bottom: Schematic illustration of the proposed mechanism for intracellular siRNA release from uPIC-AuNPs in the presence of GSH. (click on image to enlarge)
|
|
The researchers formed a polymer complex with a single siRNA molecule. The siRNA-loaded complex was then bonded to a 20 nm gold nanoparticle, which thanks to advances in synthesis techniques can be produced with a reliably low size distribution. The resulting nanoarchitecture had the optimum overall size - small enough to infiltrate cells while large enough to accumulate.
|
|
In an assay containing heparin – a biological anti-coagulant with a high negative charge density – the complex was found to release the siRNA due to electrostatic interactions. However when the gold nanoparticle was incorporated the complex remained stable. Instead, release of the siRNA from the complex with the gold nanoparticle could be triggered once inside the cell by the presence of glutathione, which is present in high concentrations in intracellular fluid. The glutathione bonded with the gold nanoparticles and the complex, detaching them from each other and leaving the siRNA prone to release.
|
|
The researchers further tested their carrier in a subcutaneous tumour model. The authors concluded that the complex bonded to the gold nanoparticle “enabled the efficient tumor accumulation of siRNA and significant in vivo gene silencing effect in the tumor, demonstrating the potential for siRNA-based cancer therapies.”
|

