| Posted: Aug 07, 2015 |
Spins in artificial atoms resemble those in natural ones
(Nanowerk News) By extending the study of coupled quantum dots to five-electron systems, RIKEN researchers have confirmed that the spin-based electron-filling rules for natural atoms apply to artificial molecules ("Vanishing current hysteresis under competing nuclear spin pumping processes in a quadruplet spin-blockaded double quantum dot").
|
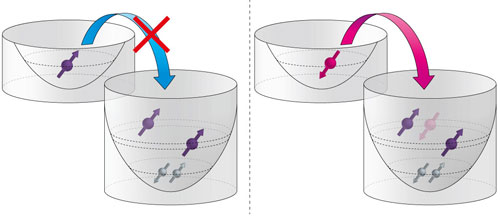 |
| Figure 1: The Pauli exclusion principle prohibits two electrons with the same spin orientation from occupying the same orbital. This prevents a spin ‘up’ electron from moving from the left quantum dot to the right one for the case shown here. (Image: Shinichi Amaha, RIKEN Center for Emergent Matter Science)
|
|
Systems consisting of electrons and semiconductor quantum dots—nanostructures that exhibit quantum properties—are highly intriguing artificial structures that in many ways mimic naturally occurring atoms. For example, electrons occupy the energy levels of quantum dots according to the same rules that determine how electrons fill atomic shells. Such systems are of both fundamental interest, for investigating phenomena related to nuclear spin, and applied interest, for manipulating spin in future quantum computers.
|
|
The Pauli exclusion principle, which prohibits any two electrons in an atom from having identical sets of quantum numbers, gives rise to a phenomenon known as the Pauli spin blockade in quantum-dot systems. This effect prevents electrons from following certain energetically favorable paths through a quantum-dot system since two electrons with the same spin cannot occupy the same energy level.
|
|
The Pauli spin blockade has been well studied in artificial molecules consisting of two quantum dots and two electrons. Shinichi Amaha and Seigo Tarucha from RIKEN’s Center for Emergent Matter Science, in collaboration with researchers in Japan and Canada, have extended the study of spin blockade to multilevel quantum-dot systems that have more than two electrons. This requires accessing high-spin states, which is difficult to achieve in practice.
|
|
Using a two-quantum-dot system with three effective levels, the researchers have achieved spin blockade by exploiting Hund’s first rule, which dictates that electrons in an atom will first fill unoccupied orbitals of a subshell with greater total spin state. They used this principle to prepare the high-spin states needed for spin blockade (Fig. 1).
|
|
The team discovered that the current of the device varied unexpectedly with the applied magnetic field. In most devices with spin effects, the current lags behind changes to the magnetic field, a phenomenon known as hysteresis. The researchers found that the hysteresis of their system follows the expected spin states based on a consideration of Hund’s rule and that in certain magnetic field regions two hysteresis effects cancelled each other out—clear evidence that competing ‘up’ and ‘down’ nuclear spin pumping processes influence the current.
|
|
These findings are expected to open the way to use arrays of such quantum dots as simulators for spin filling in real molecules. “Using an array of quantum dots as artificial atoms could assist investigations of novel spin-related phenomena in real molecules,” says Amaha.
|

