| Posted: Dec 17, 2015 |
Facile hydrolysis of the Metal-NHC framework under regular reaction conditions
(Nanowerk News) Scientists are involved in a non-stop search of an ideal catalyst, which could facilitate discovery of new reactions, as well as make known reactions more selective, environmentally friendly and economically efficient. Introduction of N-heterocyclic carbene ligands (NHC) made a real breakthrough in the catalysts’ development. In combination with transition metal, M/NHC complexes are ubiquitously utilized for developing catalytic applications in modern chemistry.
|
|
NHC complexes of transition metals are considered quite suitable for this role, as they possess unique characteristics: high activity, flexibility, tunability, outstanding diversity, and many other unique characteristics. The only “little” problem remains: failure to recycle and re-use metal/NHC complexes. Indeed, these fantastic catalysts are typically of single use and in most cases cannot be isolated back after the reaction. Since metal complexes with NHC ligands are usually considered stable, failure to recycle has been disappointing for researchers for many years.
|
|
An unusual finding was reported (Organometalics, "Facile Hydrolysis of Nickel(II) Complexes with N-Heterocyclic Carbene Ligands") by the group of researchers led by Prof. Ananikov, who noticed that Ni-NHC complexes do undergo a hydrolysis with a breakage of metal-ligand bond. Depending on the nature of the NHC ligand, noticeable decomposition takes place in the time period from minutes to hours. In some cases the complexes were even sensitive to a trace amount of water and the yield of the reaction depended on the degree of dryness of the reagents and solvents. This phenomenon prompted a question about the moisture resistance of the Ni/NHC complexes, which forced the researchers to re-think the routinely assumed picture.
|
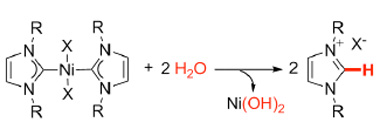 |
| Hydrolysis of the Ni-NHC complexes leading to the release of Ni hydroxide and free ligand.
|
|
The study demonstrated that the Ni/NHC complexes can either survive in aqueous solutions during the prolonged time (more than a week) or, in contrast, “live” for a couple of minutes depending on the nature of the carbene moiety. Detailed comparison of the processes showed that complexes decompose into initial reagent and nickel hydroxide. The observed speed of hydrolysis for triazolium complexes is much higher than that for imidazolium or benzimidazolium complexes.
|
|
Since water is a common contaminant in regular chemical transformations, the described phenomenon makes an impact on the catalyst’ development area Moreover, the observed degradation process may shed some light on the problem of catalyst stability and recycling.
|

