| Posted: Jan 26, 2016 |
Stable two-dimensional radioactive films: An atomic-scale picture
(Nanowerk News) Two dimensional radioactive films are a new and exciting system to study nuclear decay at the atomic level with applications in a variety of fields ranging from medical imaging to cancer therapy. Before these films can be used in real-world application however, their behaviour and stability under ambient conditions has to be understood.
|
|
A study, which was done in collaboration between the experimental group of Prof. Sykes (Tufts University, USA) and the computational group of LCN Professor, Angelos Michaelides, addresses precisely that issue.
|
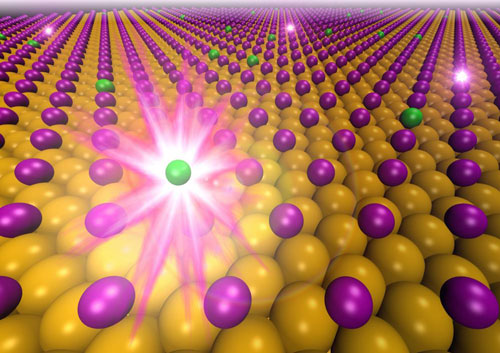 |
| Nuclear transmutation of 125I to 125Te (green). Violet spheres represent the radioactive iodine atoms adsorbed on a gold (111) surface.
|
|
Using X-ray photoelectron spectroscopy and scanning tunneling microscope experiments combined with density functional theory computations the authors were able to monitor the decay product, 125Te, over time with atomistic resolution. This work reveals not only that the radioactive films and the decay product are stable in air at ambient conditions, but also shows precisely what happens to these films over time.
|
|
Freshly formed Te is bound very strongly to the gold substrate, even stronger than the radioactive iodine atoms, and oxidises to TeO2 in air. TeO2 units are able to diffuse through the films and tend to dimerize to (TeO2)2. The radioactive films as well as the decay products remain intact throughout these reactions. This crucial insight opens the door for a range of useful applications of two dimensional radioactive films on gold. Adsorbed on gold nanoparticles they could for example lead to highly targeted cancer therapy treatments.
|
|
This study builds upon previous collaborative work, published in Nature Materials, in which the radioactive films were been introduced to the community and is available to download in ACS Nano ("Atomic-Scale Picture of the Composition, Decay, and Oxidation of Two-Dimensional Radioactive Films").
|

