| Posted: May 20, 2016 |
Right size + right chemistry = right stuff for plastics manufacturing
(Nanowerk News) Plastic manufacturing is an energy-intensive process. Now, research performed in part at the National Institute of Standards and Technology (NIST) has revealed a way to reduce the energy demand in one key step of plastic manufacturing by using a class of materials that can filter impurities more efficiently than the conventional manufacturing process.
|
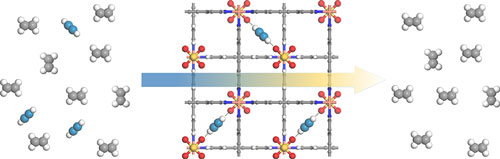 |
| Ethylene (on left in gray) is usually contaminated with acetylene (blue), which can ruin the process that creates the polyethylene used in most plastic. SIFSIX metal-organic frameworks (center) can capture the acetylene efficiently, leaving pure ethylene (right). (Image: Zhou/NIST) (click on image to enlarge)
|
|
The findings, published in the journal Science ("Pore chemistry and size control in hybrid porous materials for acetylene capture from ethylene"), show that materials called metal-organic frameworks (MOFs) can effectively remove the contaminant acetylene from ethylene, the material from which much of the world’s plastic is made. The research suggests that filtering out acetylene using MOFs would produce ethylene at the high purity that industry demands while sidestepping the current need to convert acetylene to ethylene via a costly catalytic process.
|
|
The chemical name for the plastic you see every day—from water bottles and grocery bags to household appliances—is polyethylene, a pliable material made by stringing together long chains of a simpler molecule called ethylene. Worldwide demand for plastic makes ethylene the most widely produced organic compound in the world, with well over 100 million tons of it manufactured each year, largely by refining crude oil.
|
|
Newly made ethylene is not pure enough to become plastic because the refinement process also creates a substantial amount of acetylene, which can ruin the catalysts that enable ethylene molecules to be strung together. The conventional industrial solution is to convert this undesirable acetylene into ethylene as well, but this step requires the use of palladium, an expensive and rare metal, as a catalyst and consumes a significant amount of energy.
|
|
The research team, which includes scientists from the NIST Center for Neutron Research (NCNR) and five universities from around the world, found that a family of MOF materials called SIFSIX, discovered in the 1990s, might provide a better alternative for removing the acetylene. MOFs are porous crystals that under a microscope look a bit like a building under construction—lots of girders with space in between. The SIFSIX group gets its name from some of its girders, which are formed from silicon (Si) and six atoms of fluorine (F6).
|
|
The team found that when they passed ethylene through the MOFs, the fluorine attracted and captured most of the acetylene contaminant, letting the now-purified ethylene to pass unhindered. Varying the size of the pores by changing the length of the girders allowed the MOFs to filter ethylene-containing acetylene in concentrations of anywhere from 1 percent to 50 percent, which are typical in industry.
|
|
The SIFSIX MOFs set records among adsorbent materials for both selectivity (the ability to attract the acetylene only while allowing the ethylene to pass) and adsorption capacity. According to the research team, the results show that the SIFSIX group offers a viable alternative to standard industrial practice.
|
|
“They reduced the amount of acetylene in ethylene down to less than 2 parts per million (ppm), which is lower than the 5 ppm that polyethylene manufacturing requires,” said NIST materials scientist Wei Zhou. “SIFSIX MOFs are easy to produce, safe to use, and can be reused over and over again. They also have the advantage of being stable, which is not true of all MOFs.”
|
|
The MOFs were created and investigated in great detail by researchers based at China’s Zhejiang University (by Huabin Xing), Ireland’s University of Limerick, (Michael Zaworotko) and the University of Texas–San Antonio in the U.S. UT-San Antonio Professor Banglin Chen sensed the significance of SIFSIX MOFs for this application, and organized and led the team. The NIST portion of the work, which involved computer modeling of the MOFs and neutron diffraction experiments, clarified the mechanism by which the SIFSIX MOFs captured the acetylene. Scientists from the Netherlands’ University of Amsterdam and Saudi Arabia’s King Abdullah University of Science and Technology also contributed.
|

