| Posted: Aug 09, 2016 |
Engineering a chemical switch into a light-driven proton pump
(Nanowerk News) Synthetic biology is an emerging and rapidly evolving engineering discipline. Within the NCCR Molecular Systems Engineering, Bernese scientists have engineered a chemically switchable version of the light-driven proton pump proteorhodopsin – an essential tool for efficiently powering molecular factories and synthetic cells.
|
|
Synthetic biology is a highly interdisciplinary field, which combines biology, chemistry and physics with engineering. Its goal is to design molecular factories and synthetic cells with novel properties or functions for applications in healthcare, industry, or biological and medical research. Such artificial systems are in the nanometer scale and are built by combining and assembling existing, synthetic or engineered building blocks (e.g., proteins). Molecular systems have wide application ranges, e.g., for chemical compound synthesis, waste disposal, energy supply and medical diagnosis or treatment.
|
|
In this context, the NCCR Molecular Systems Engineering brings Swiss scientists from different disciplines together to stimulate innovation, and address existing and future challenges. The University of Bern is represented by the Fotiadis laboratory in the NCCR MSE.
|
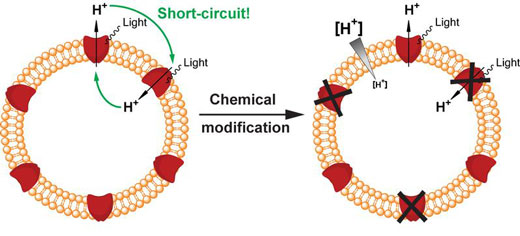 |
| Nanocontainer consisting of lipid (in orange) and the engineered version of proteorhodopsin (in red). Light irradiation of the molecular system fails in establishing a proton gradient across the membrane because of the symmetrical integration of proteorhodopsins, which results in a short-circuit (left). After chemically switching off wrongly oriented proteorhodopsins (right), correctly oriented ones can establish a proton gradient across the membrane (indicated in gray). (Image: Dimitrios Fotiadis, University of Bern)
|
|
Nanomachines for energy conversion
|
|
Energy-providing building blocks are essential to power molecular systems. Light-driven proton pumps such as the membrane protein proteorhodopsin represent excellent nanomachines for efficient energy conversion. Light energy, e.g., solar energy, is easily accessible and efficiently used by proteorhodopsin to establish proton gradients across membranes, which separate two different compartments. Such gradients can then be used to drive proton-driven building blocks of molecular systems, for example proton-driven transporters. Living cells commonly use proton gradients to power processes such as import and export of solutes and ions through transporters, and the synthesis of metabolites.
|
|
Eliminating the short-circuit
|
|
Using common methods for the assembly of proteorhodopsin, and membrane proteins in general, into containers such as liposomes or polymerosomes (i.e., spherical structures consisting of lipid or polymer membranes), symmetric integration in membranes is observed leading to short-circuit and failure in establishing a proton gradient. Therefore, members from the Fotiadis group, in particular Dr. Daniel Harder and Stephan Hirschi, together with colleagues from the NCCR MSE have implemented a chemical on-off switch into proteorhodopsin, thus extending its versatility and allowing the establishment of an asymmetric distribution of functional proteorhodopsin proteins in membranes by selectively deactivating one of the two possible orientations.
|
|
This engineered version of proteorhodopsin represents the first light-driven proton pump and energizing-building block that can be activated and deactivated chemically to meet the requirements of the molecular system. «Possible applications of this versatile energy-providing building block in specific molecular factories represent the light- and solar-powering of the production of molecules such as life’s universal energy currency ATP (adenosine triphosphate) and of the bioremediation of pollutants such as antibiotics in water resources», says Fotiadis. The study was published in the renowned scientific journal Angewandte Chemie International Edition ("Engineering a Chemical Switch into the Light-driven Proton Pump Proteorhodopsin by Cysteine Mutagenesis and Thiol Modification").
|

