| Posted: Oct 06, 2016 |
New efficient, low-cost method to hydrogenate graphene with visible light
(Nanowerk News) An environmentally friendly, efficient and low-cost method for hydrogenation of graphene with visible light has been developed by researchers at Uppsala University and AstraZeneca Gothenburg, Sweden. The research study is presented in an article in Nature Communications An environmentally friendly, efficient and low-cost method for hydrogenation of graphene with visible light has been developed by researchers at Uppsala University and AstraZeneca Gothenburg, Sweden. The research study is presented in an article in Nature Communications ("Metal-Free Photochemical Silylations and Transfer Hydrogenations of Benzenoid Hydrocarbons and Graphene").
|
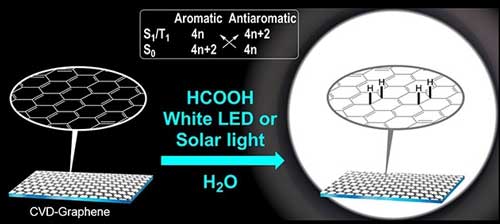 |
|
The study shows that the two-dimensional and atom-thin carbon material graphene reacts with formic acid in a water solution upon irradiation with visible light. In the reaction, formic acid acts as masked hydrogen and a material is produced where hydrogen extensively has been added to graphene. One says that graphene has been hydrogenated. The study was performed by Assoc. Prof. Henrik Ottosson’s research group at the Department of Chemistry – Ångström Laboratory, together with colleagues in Chemistry, Physics and Engineering at Uppsala University and at AstraZeneca Gothenburg.
|
|
“The reaction is convenient and cheap, and hydrogenated graphene may be applied within areas such as hydrogen storage. Additionally, upon functionalization of graphene one can open a band gap and this fact is of high relevance for electronics applications”, says Henrik Ottosson.
|
|
Yet, graphene research is a side-project in Henrik Ottosson’s group. The group normally studies the behaviours of various aromatic hydrocarbons upon irradiation, and they apply a rule, the so-called Baird’s rule, which can be derived through chemically applied quantum mechanics.
|
|
Chemical compounds that are aromatic have an inherently high stability and often they are not easy to degrade. Benzene is the most well known aromatic compound and more than half of all known chemical compounds contain aromatic groups.
|
|
The high stability of aromatic compounds is explained by Hückel’s ‘4n+2’ rule, but this rule is only valid for compounds in their electronic ground states. Upon exposure to light of a certain wavelength, the aromatic compounds reach electronically excited states. According to Baird, compounds that are aromatic in the ground state become antiaromatic and reactive in the excited state. The rule, neglected for decades, can now be used to describe various behaviours of aromatic compounds when irradiated.
|
|
Using Baird’s rule, Henrik Ottosson’s group develops new light-initiated reactions. First, they studied addition of hydrosilanes to benzenes, naphthalene and gradually larger polycyclic aromatic hydrocarbons (hydrosilanes are compounds that can be regarded as heavy analogues of hydrogen). Despite the fact that it is not possible to explain if, and how, Baird’s rule can be applied to graphene (an essentially infinitely large polycyclic aromatic hydrocarbon), the group explored graphene chemistry and found a very efficient addition reaction when using formic acid.
|

